1. Overview
rcisignal consolidates the quality-assurance workflow
for reverse-correlation (RC) experiments into one toolkit. It addresses
three questions, in order. First, are the inputs clean (response coding,
trial counts, response bias, stimulus- pool alignment)? Second, is the
signal informative and stable (does each condition’s group CI carry more
pattern than chance, and would the pattern replicate on a different half
of the producers)? Third, when there is more than one condition, are the
conditions distinguishable, both in overall magnitude and in spatial
location?
Two halves of the package address these questions in turn. The
input-side diagnostics (run_diagnostics() and the
check_* family) cover the first question. The output-side
reliability and discriminability metrics
(run_reliability(), run_discriminability(),
infoval(), agreement_map_test(), together with
the lower-level building blocks rel_*() and
pixel_t_test()) cover the second and third.
1.1 Scope
For 2IFC stimulus generation and CI computation,
rcisignal delegates to the upstream rcicr package
(Dotsch, 2016, 2023). ci_from_responses_2ifc() is a small
convenience function around rcicr::batchGenerateCI2IFC()
that takes care of the integration quirks. Brief-RC support (Schmitz,
Rougier, & Yzerbyt, 2024) is built into rcisignal directly, because
rcicr does not yet provide it.
The metrics in this package quantify whether a CI is stable (within-condition) and separable (between-condition). Whether the CI accurately reflects the producer’s mental representation of the target trait is a separate validity question, typically addressed by an external rater study, and sits outside the package. Cone, Brown-Iannuzzi, Lei, & Dotsch (2021) showed that the standard two-phase rating design inflates Type I error; rcisignal’s metrics operate directly on producer-level pixel signal and thereby sidestep that pitfall.
The intended audience is RC researchers at an intermediate R level
with basic familiarity with the rcicr package or with the
Schmitz et al. (2024) Brief-RC structure. No prior expertise in
data.table, permutation testing, or intraclass correlation
is assumed.
1.2 Validation status
Worth flagging before any published use of this package: not all of the metrics it ships are independently validated for social-face RC data. The package is best treated as a toolbox that collects existing methods, plus a few natural extensions of those methods, into one place. Some of those extensions are mature and well-grounded in adjacent literatures; others are sensible-looking implementations that have not yet had a dedicated validation study on the kind of data this package targets. Reporting accordingly matters.
Validated in their respective domains:
-
Per-producer infoVal for 2IFC (Brinkman et al.,
2019). The z-score derivation, reference distribution, and the
z >= 1.96threshold all originate from the Brinkman et al. paper, which validated the metric on social-face 2IFC data. - Pixel-test methodology (Chauvin et al., 2005). The Welch-style per-pixel t-statistic, plus its smoothing and inferential framing, are well-established in the classification-image and neuroimaging literatures.
- Cluster-based permutation tests for FWER control (Maris & Oostenveld, 2007). Validated on EEG and MEG data, with the underlying logic carrying over to any spatial statistical map.
- Threshold-free cluster enhancement (TFCE) (Smith & Nichols, 2009). Validated on neuroimaging data; same transferability caveat as above.
Package-level extensions, not yet independently validated for social-face RC:
- Group-mean infoVal. A natural extension of per-producer infoVal to the group-average CI, with a trial-count-matched reference. Brinkman et al. (2019) recommend reporting the distribution of per-producer infoVals rather than a single group z; the group-mean version is offered here as a supplementary summary, not as a replacement for the per- producer reporting.
- Between-condition discriminability tests (cluster-based permutation and TFCE). The underlying machinery is borrowed from neuroimaging where it is well-validated; its specific behaviour on social-face CI maps (which differ from EEG/MEG or fMRI in spatial structure, signal-to-noise, and base-image artefacts) has not been the subject of a dedicated validation study.
-
Pixel-wise agreement / reliability maps
(
agreement_map_test()and the related plot helpers). Same machinery as above, applied within a single condition. Same caveat. - infoVal applied to Brief-RC. The Frobenius-norm logic transfers, and the trial-count-matched reference closes the most obvious calibration gap relative to a pool-keyed reference. The threshold conventions inherited from 2IFC have not been re-validated on Brief-RC.
If you use the unvalidated metrics in published work, please report them as exploratory and indicate the package version. If you are aware of validation studies I have missed, I would be glad to update this section (m.j.barbosa.de.oliveira@tue.nl).
2. Installation
# Latest release from GitHub.
remotes::install_github("olivethree/rcisignal",
dependencies = TRUE)
# rcicr is a Suggests dep; install it if you need the 2IFC path.
install.packages("rcicr") # CRAN
remotes::install_github("rdotsch/rcicr") # developmentThe mandatory dependencies are minimal (cli and
data.table, plus the base packages). PNG and JPEG readers
(png, jpeg), rcicr for 2IFC
pipelines, and psych for ICC cross-validation sit in
Suggests and load on demand.
rcisignal is in an experimental stage and exported
functions are still being refined. Re-running the
install_github() call above at the start of each analysis
session pulls the latest version; this user guide is kept in sync with
new and updated functions.
2.1 Quickstart with simulated data
Two helpers, simulate_2ifc_data() and
simulate_briefrc_data(), generate a complete synthetic
dataset (responses + noise pool +, for 2IFC, an
rcicr-format .Rdata) so the rest of this
vignette can be exercised without needing to bring your own files. They
are also building blocks for simulation studies (power, calibration of
reliability and discriminability metrics, sensitivity to
contamination).
What they generate
-
Responses. A long-format
data.tablewith one row per trial and the columns every diagnostic / CI function expects:participant_id,condition,trial,stimulus,response(in{-1, +1}),rt(in milliseconds). -
Noise pool. A
pixels x n_trialsnumeric matrix, generated on the fly viarcicr::generateNoisePattern()andrcicr::generateNoiseImage(). (rcicrmust be installed; with the default 256-pixel images and 500 trials the pool takes roughly one to three minutes, with acliprogress bar.) -
For 2IFC, an
.Rdatafile in the format thatrcicr::generateStimuli2IFC()writes, soci_from_responses_2ifc(),compute_infoval_summary(), and every other function that asks for anrdataargument works out of the box. -
A self-contained
$stimulilist that round-trips throughsaveRDS()/readRDS()and a$base_image_pathPNG written next to the stimuli.Rdata. The first survives session restarts when handed to a consumer viastimuli =; the second is the rcicr-style on-disk artefact for tools that expect a base-face file.
The return value is an rcisignal_sim S3 object:
sim <- simulate_2ifc_data()
str(sim, max.level = 1)
#> List of 10
#> $ data : data.table [50000 x 6]
#> $ noise_matrix : num [1:65536, 1:500] (pixels x trials)
#> $ base_face : num [1:256, 1:256]
#> $ params : num [1:500, 1:4092] (rcicr stimuli_params)
#> $ p : list of 4 (rcicr noise basis)
#> $ signal : num [1:65536] (planted signal vector)
#> $ rdata_path : chr "/tmp/.../rcisignal_sim_2ifc_stimuli.Rdata"
#> $ base_image_path : chr "/tmp/.../rcisignal_sim_2ifc_base_face.png"
#> $ stimuli : list of 11 (portable, round-trips via saveRDS)
#> $ meta : list (seed, elapsed, etc.)Defaults at a glance
| Argument | Default | Notes |
|---|---|---|
n_per_condition |
50 |
participants per condition |
conditions |
c("target", "control") |
any character vector works |
n_trials |
500 |
per participant; equals noise pool size for 2IFC |
images_per_trial (Brief-RC only) |
12 |
= 6 original/inverted pairs |
noise_pool_size (Brief-RC only) |
n_trials * (images_per_trial / 2) |
shared across participants |
img_size |
256 |
pixels; matches the bundled base face |
base_image |
inst/extdata/sim_base_face.png |
a 256x256 grayscale artificial face; pass a path or matrix to override |
signal_strength |
"weak" |
also "none" (true null), "strong", or a
numeric coefficient |
signal_region |
"eyes" |
any region accepted by make_face_mask()
|
rt_contamination_fast / _slow
|
0.02 / 0.02
|
fraction of trials replaced by uniform-fast (50-200 ms) / uniform-slow (5000-20000 ms) responses |
noise_type, nscales,
sigma
|
"sinusoid", 5, 25
|
forwarded to rcicr::generateNoisePattern()
|
rdata_dir |
NULL |
optional directory for a stable-path stimuli .Rdata;
pass to keep the sim usable across R sessions |
seed |
NULL |
a random seed is drawn and stored on the result |
progress |
TRUE |
shows a cli progress bar during noise generation |
Signal model
Each trial’s response is drawn from a logistic / softmax model whose
location depends on a planted pixel-level signal s (the
binary mask returned by make_face_mask() for the chosen
signal_region).
-
2IFC. On each trial
tthe participant sees image_a = base + noise[t] and image_b = base - noise[t] and chooses one. The log-odds of choosing image_a (response =+1) arebeta * (noise[, t] %*% s) / sqrt(sum(s)). Withsignal_strength = "none"(beta = 0), choices are uniform random; with"weak"(beta = 0.5) the planted region biases responses just enough that a 50 x 2 x 500 dataset yields a recognisable cluster on the eyes region;"strong"(beta = 2) produces a much sharper signal. -
Brief-RC. Each trial shows
images_per_trial = 2kimages (the original and inverted versions ofkdistinct noise patterns drawn from the shared pool). Each image gets a Gumbel-perturbed utility±beta * (noise %*% s) / sqrt(sum(s)) + Gumbel(0,1)and the participant picks the argmax (multinomial-logit / softmax). The recordedstimulusis the pool index of the chosen pair;responseis+1if the original version of that pair was chosen,-1if the inverted version.
A weak signal is the default rather than "none" so the
worked example produces a recognisable CI on the planted region rather
than a flat null result. Pass signal_strength = "none" to
get truly bogus data (useful for testing the diagnostic side,
calibrating null distributions, or stress-testing the reliability /
cluster permutation code under no-signal conditions).
Response-time model
RTs follow a shifted lognormal
(rt = round(exp(rnorm(n, log(800), 0.5)) + 150), in
milliseconds) with two contaminant streams:
-
Fast contaminants at
rt_contamination_fast(default 2%): uniform[50, 200]ms, mimicking accidental clicks. -
Slow contaminants at
rt_contamination_slow(default 2%): uniform[5000, 20000]ms, mimicking distraction or task pauses.
These are deliberately tuned so that check_rt() finds
something to flag (useful for sanity-checking the RT diagnostic without
curating real outliers by hand).
End-to-end demo (2IFC)
Pasting the chunk below into a fresh R session takes you from no data at all to a within-condition reliability summary:
sim <- simulate_2ifc_data(
n_per_condition = 30, # smaller for a quick demo
n_trials = 200,
signal_strength = "weak",
seed = 1
)
# Step 1: run the diagnostic battery on the simulated responses.
# Pass the simulator's rdata so rdata-dependent sub-checks (response
# inversion, infoval consistency) run too. `stimuli = sim$stimuli`
# is an in-memory equivalent.
print(run_diagnostics(sim$data, method = "2ifc",
rdata = sim$rdata_path, col_rt = "rt"))
# Step 2: compute per-participant CIs using the bundled .Rdata.
target_rows <- subset(sim$data, condition == "target")
control_rows <- subset(sim$data, condition == "control")
cis_target <- ci_from_responses_2ifc(target_rows,
rdata_path = sim$rdata_path)
cis_control <- ci_from_responses_2ifc(control_rows,
rdata_path = sim$rdata_path)
# Step 3: within-condition reliability.
print(run_reliability(cis_target$signal_matrix, seed = 1))
print(run_reliability(cis_control$signal_matrix, seed = 1))
# Step 4: between-condition cluster test.
print(run_discriminability(
signal_matrix_a = cis_target$signal_matrix,
signal_matrix_b = cis_control$signal_matrix,
seed = 1
))For the Brief-RC pipeline the equivalent demo replaces
simulate_2ifc_data() with
simulate_briefrc_data() and
ci_from_responses_2ifc() with
ci_from_responses_briefrc(). The Brief-RC consumer reads
the noise matrix directly, so the call becomes
ci_from_responses_briefrc(sim$data, noise_matrix = sim$noise_matrix);
add base_image = sim$base_face if you also want the
rendered visualisation (scaling = "matched").
A note on speed
Noise generation is the dominant cost (about 0.4-0.5 s per trial at
256 pixels with default basis settings, in pure R). With default
parameters expect roughly 1-3 minutes per call. A future release may
provide an Rcpp accelerator; for now the function is
intentionally single-shot (“generate once, reuse the
rcisignal_sim object across many analyses”).
To pay the cost only once across R sessions (saveRDS() /
readRDS(), knitr cache = TRUE, sharing with a
collaborator), use one of two portable routes. Pass
rdata_dir = "simdata/" to the simulator so the stimuli
.Rdata keeps a stable path, or hand
stimuli = sim$stimuli to the consumer in place of
rdata_path = sim$rdata_path. The $stimuli list
is self-contained and survives session restarts even after the
.Rdata file is gone.
3. Signal matrix
Almost every analytical function in rcisignal operates
on a single data structure: a signal matrix with one
row per pixel and one column per producer (participant). The two
top-level functions run_reliability() and
run_discriminability() take a signal matrix as input, and
so do the lower-level rel_split_half(),
rel_icc(), rel_loo(),
pixel_t_test(), rel_cluster_test(),
rel_dissimilarity(), infoval(), and
agreement_map_test(). Once you have the signal matrix in
the right shape, the rest of the analysis follows.
3.0 Three pixel matrices that all sound similar: keep them apart
Reverse correlation work involves several types of pixel matrices
that may be easy to confuse. In rcisignal, each one has
exactly one job:
| Data type | What is it? | shape | Where it comes from |
|---|---|---|---|
noise_matrix |
input pool of noise patterns the experiment chose stimuli from. One column per pre-generated noise pattern. |
n_pixels × pool_size
|
input (you give it to the package) |
| noise mask (a.k.a. “per-participant CI”) | one participant’s classification image: a single vector of pixel values, base-subtracted. Conceptually, the weighted average of the noise patterns they “selected” with their responses. |
n_pixels × 1 (one column) |
intermediate |
signal_matrix |
all participants’ noise masks stacked side by side,
one column per producer. This is the central object of
rcisignal. |
n_pixels × n_participants
|
output (you pass it to every rel_*,
run_reliability, run_discriminability
call) |
In sum:
-
noise_matrixdescribes the stimuli (what the experiment showed), and exists before any participant runs the task (see §4.3 for how to read it); - noise mask describes one participant’s CI, and exists after that participant has responded;
-
signal_matrixdescribes the whole sample’s CIs. It is the collection of all individual noise masks lined up by participant (this is the matrix every reliability and discriminability function inrcisignaluses).
You don’t build the noise mask or the signal_matrix by
hand: ci_from_responses_2ifc() and
ci_from_responses_briefrc() do it for you and return a list
whose $signal_matrix element is the matrix you pass to the
metrics in §7-§9.
A small terminology trap. The word mask above means image-shaped overlay (one number per pixel, defined over the whole image grid). It is not the same as a face-region mask (a logical 1/0 stencil that selects “eyes” or “mouth” pixels); those are covered separately in §4.5.
A note on the signal matrix name. Other RC papers sometimes
call this same per-producer object a noise matrix, because the
underlying pixel values are visual noise patterns. Both names are
reasonable: the data really do contain a mixture of noise (the per-trial
random patterns the experiment showed) and signal (the producer’s
sign-weighted aggregation of those patterns). The metrics in this
package are designed to test how much of that mixture is signal rather
than noise, hence signal matrix. To avoid the name collision,
rcisignal’s code reserves noise_matrix
strictly for the input pool above (the row of the table) and
signal_matrix strictly for the per-producer output (the
third row). Whatever you call the object in your own writing, the shape
and interpretation are the same.
Two paths lead to a signal matrix, with different consequences for the metrics that follow.
3.1 Two paths to the signal matrix
Mode 2: from raw trial-level responses
(recommended). Use ci_from_responses_2ifc() for
2IFC pipelines or ci_from_responses_briefrc() for Brief-RC.
Both return a list with $signal_matrix already in the right
shape, base-subtracted, and unscaled (i.e. carrying the raw mask). This
is the safe path for the reliability metrics later on.
res <- ci_from_responses_2ifc(
responses,
rdata_path = "data/rcicr_stimuli.Rdata",
baseimage = "base"
)
signal <- res$signal_matrixMode 1: from pre-rendered CI PNGs on disk. Use
read_cis() to read a directory of PNG/JPEG CIs, followed by
extract_signal() (or the read_signal_matrix()
shortcut that composes both). This path is offered for convenience and
carries a caveat: PNG pixels are necessarily what was rendered to disk
(base + scaling(mask)). After base subtraction, the
resulting signal is scaling(mask) rather than the raw
mask.
signal <- read_signal_matrix(
dir = "data/cis_condition_A/",
base_image_path = "data/base.jpg"
)3.2 Raw mask vs rendered CI
For correlation-based metrics (rel_split_half(),
rel_loo()), the rendered scaling is mostly harmless because
a single uniform linear stretch preserves Pearson correlation. For
variance-based metrics (rel_icc(),
pixel_t_test(), the cluster test, and the Euclidean half of
rel_dissimilarity()), scaling distorts the numbers. The
"matched" (per-CI) scaling option, where each producer’s
mask is stretched to the base’s dynamic range, breaks correlation-based
metrics as well.
rcisignal keeps track of which kind of matrix you have
by labelling each signal matrix with a source tag (either
"raw" or "rendered"). The variance-based
functions check this tag before they run, and stop with an informative
error if you pass a rendered matrix:
- Functions that build raw masks
(
ci_from_responses_2ifc(),ci_from_responses_briefrc()) label the resulting matrix as"raw". - Functions that read PNGs (
read_cis(),extract_signal(),read_signal_matrix()) label it as"rendered". - Variance-based metrics check this label internally and stop with an
informative error when given a
"rendered"matrix, unless you passacknowledge_scaling = TRUEto confirm you have read the caveat.
# This works:
rel_icc(res$signal_matrix)
# This errors with a clear message:
rel_icc(read_signal_matrix("cis/", "base.jpg"))
#> Error: signal_matrix is a rendered CI (PNG-derived); ...
# Override after reading the explanation:
rel_icc(read_signal_matrix("cis/", "base.jpg"),
acknowledge_scaling = TRUE)A safety check (looks_scaled()) also flags hand-built
signal matrices that don’t carry the source label but whose
value range looks rescaled. This check emits a once-per-session warning
rather than stopping the analysis; silence it with
options(rcisignal.silence_scaling_warning = TRUE).
One important exception: rcicr::computeInfoVal2IFC() is
unaffected by display scaling. It reads the raw $ci element
from the rcicr CI list internally
(norm(matrix(target_ci[["ci"]]), "f")) regardless of the
scaling argument used at generation, so the standard 2IFC
infoVal path is safe even when the displayed CIs are rendered.
Hand-rolled implementations (including
rcisignal::infoval(), which has to support Brief-RC where
no upstream function exists) require the raw mask explicitly.
4. Data preparation
This section covers the four objects the package consumes: trial-level responses, the noise matrix, a base image, and an optional face mask.
4.1 Response data
Trial-level data, one row per trial, in any tabular shape
(data.frame, data.table, tibble).
Required columns:
| Column | Type | Meaning |
|---|---|---|
participant_id |
char/int | producer identifier |
stimulus |
int | stimulus / pool id (range depends on method, see below) |
response |
+1 / -1
|
producer’s choice (see below) |
rt (optional) |
numeric | response time in ms (needed only for check_rt()) |
2IFC response coding
Each trial presents two faces drawn from a unique noise pair.
response = +1 if the producer picked the original variant
(base + noise), and -1 if they picked the
inverted variant (base - noise). A common silent failure in
2IFC pipelines is {0, 1} coding produced by experiment
software that records “left” / “right” as 0 / 1;
check_response_coding() flags this with a recode formula in
the suggestion text.
A 2IFC dataset with three participants and four trials each illustrates the format. On every trial the participant saw two stimuli (one original and one inverted noise pattern superimposed on the same base face) and chose one:
responses_2ifc <- data.frame(
participant_id = rep(c("P01", "P02", "P03"), each = 4),
stimulus = rep(1:4, times = 3),
response = c( 1, -1, 1, 1,
-1, 1, 1, -1,
1, 1, -1, 1),
rt = c(820, 910, 750, 880,
680, 1040, 720, 950,
900, 770, 990, 810)
)
responses_2ifc
#> participant_id stimulus response rt
#> 1 P01 1 1 820
#> 2 P01 2 -1 910
#> 3 P01 3 1 750
#> 4 P01 4 1 880
#> 5 P02 1 -1 680
#> 6 P02 2 1 1040
#> 7 P02 3 1 720
#> 8 P02 4 -1 950
#> 9 P03 1 1 900
#> 10 P03 2 1 770
#> 11 P03 3 -1 990
#> 12 P03 4 1 810The 2IFC stimulus column indexes the trial’s
stimulus pair, so its range is 1:n_trials. Every trial
has its own unique pair, so an id never repeats across trials within a
participant.
Brief-RC response coding (Schmitz et al. 2024)
Each trial presents 2k noisy faces (k
original images, base + noise_i, and k
inverted images, base - noise_i), drawn from k
distinct pool noise patterns. The producer picks one. The data records
one row per trial: stimulus = pool id of
the chosen noise pattern; response = +1 if original chosen,
-1 if inverted. Unselected faces are absent from the data;
do not pad them as zero rows. The same row format applies to both
validated Brief-RC variants (Brief-RC 12 with k = 6,
Brief-RC 20 with k = 10); the analysis pipeline is
identical (see §13.1 for the formula being symmetric in
k).
A Brief-RC 12 dataset with the same three participants and four trials each illustrates the format:
responses_briefrc <- data.frame(
participant_id = rep(c("P01", "P02", "P03"), each = 4),
stimulus = c( 47, 112, 8, 263,
91, 17, 204, 55,
188, 142, 261, 73),
response = c( 1, -1, 1, 1,
-1, 1, -1, 1,
1, 1, -1, -1),
rt = c(1100, 1340, 980, 1210,
890, 1450, 1020, 1130,
1280, 1190, 1360, 1080)
)
responses_briefrc
#> participant_id stimulus response rt
#> 1 P01 47 1 1100
#> 2 P01 112 -1 1340
#> 3 P01 8 1 980
#> 4 P01 263 1 1210
#> 5 P02 91 -1 890
#> 6 P02 17 1 1450
#> 7 P02 204 -1 1020
#> 8 P02 55 1 1130
#> 9 P03 188 1 1280
#> 10 P03 142 1 1190
#> 11 P03 261 -1 1360
#> 12 P03 73 -1 1080What pool_size means concretely
In Brief-RC the stimulus column ranges from
1 to pool_size, where pool_size
is the total number of distinct noise patterns generated for the
experiment, i.e., the number of columns in the
noise_matrix (§4.3). On every trial the software draws 6
distinct pool patterns and presents each in both original and inverted
form, giving 12 alternatives. Across many trials, the same pool id can
therefore re-appear (and a producer can pick the same pool id more than
once). The exact re-use rate depends on the experimenter’s sampling
design, of which three regimes are common.
-
Without replacement at the presentation level: the
only path open when
n_trials x stim_per_trial == pool_size. Each pool item is shown exactly once across the whole task. A producer cannot choose the same pool id twice. Schmitz et al.- Experiment 1 used this regime (60 trials x 12 alternatives = 720
presentations, exactly matching their
pool_size = 720).
- Experiment 1 used this regime (60 trials x 12 alternatives = 720
presentations, exactly matching their
-
With replacement at the presentation level:
required when
n_trials x stim_per_trial > pool_size. Pool items are drawn randomly with possible repetition. A producer can choose the same pool id on two different trials (with the same response sign or with opposite signs). Example: 300 trials x 12 alternatives = 3600 presentations drawn from a 1500-item pool. - Hybrid designs (partial blocks, Latin squares, counterbalanced subsets per condition). Treat as with-replacement at the analysis level unless your design guarantees no repetition.
rcisignal is agnostic to the regime. Internally, before
computing the per-producer mask, it collapses any duplicated
stimulus ids in a producer’s data using
mean(response) exactly as Schmitz’s genMask()
formulation does. So if the same pool item is chosen twice with the same
sign, it contributes once with full weight; if chosen twice with
opposite signs, the two cancel and it contributes zero. The
genMask() divisor is
length(unique(chosen_stimuli)), not
n_trials.
Structural differences between 2IFC and Brief-RC data
| Aspect | 2IFC | Brief-RC 12 |
|---|---|---|
| Alternatives shown per trial | 2 (one original + one inverted) | 12 (six original + six inverted, drawn from six pool patterns) |
| Rows recorded per trial | 1 | 1 |
What stimulus indexes |
The trial’s stimulus pair | The chosen pool item only |
Range of stimulus
|
1 to n_trials
|
1 to pool_size
|
| Same id can repeat across trials | No (each trial has its own pair) | Depends on the experimenter’s sampling design (see above) |
| Unchosen alternatives recorded | Not applicable (only two shown) | No (treated as absent; do not pad as zero rows) |
read_responses() is a small wrapper around
data.table::fread() that validates the required
columns:
responses <- read_responses("study1data.csv", method = "2ifc")4.2 The .RData from
rcicr::generateStimuli2IFC()
The 2IFC pipeline uses an .RData file produced by
rcicr::generateStimuli2IFC(). The objects in this file that
the analysis actually uses are:
-
base_faces: the loaded base-face matrices, grayscale in[0, 1]. List names (e.g."base") become thebaseimageargument used by later functions.base_face_filescarries the matching source paths. -
img_size: side length of the (square) image in pixels. -
p: the noise basis (with$patchesand$patchIdx), the sinusoidal dictionary used to assemble each trial’s noise. -
stimuli_params: a named list of matrices (one per base label) where each row carries one trial’s contrast weights. Reconstruct triali’s noise viarcicr::generateNoiseImage(stimuli_params[[base]][i, ], p).
n_trials, seed, label,
stimulus_path, trial,
generator_version, and use_same_parameters are
bookkeeping fields, not consumed by analysis.
reference_norms is created and inserted in place by
rcicr::computeInfoVal2IFC() on its first call; copy the
rdata first if you want it untouched.
The actual per-trial noise patterns are not stored
in the rdata. They are reconstructed on demand from
stimuli_params and p;
rcisignal::read_noise_matrix() does this automatically
(§4.3) and caches the result.
On macOS the file is saved with a lowercase .Rdata
extension; list.files(pattern = "\\.RData$") is
case-sensitive by default and will miss it. Use
ignore.case = TRUE when searching.
4.3 The noise matrix
The noise matrix is an n_pixels x pool_size numeric
matrix where each column is the noise pattern shown for one trial (or
pool item). It serves as input to CI computation, distinct from the
signal matrix, which is an output.
read_noise_matrix() is a single entry point that detects
the file format automatically. For formats that are slow to parse, it
saves a faster .rds copy next to the original and re-uses
it on subsequent calls:
# Plain text matrix (Schmitz et al. 2024 OSF format).
# First call parses + writes data/noise_matrix.rds.
nm <- read_noise_matrix("data/noise_matrix.txt")
# Second call loads from the cache (fast).
nm <- read_noise_matrix("data/noise_matrix.txt")
# rcicr .Rdata source: reconstructs each trial via
# rcicr::generateNoiseImage() and caches the result.
nm <- read_noise_matrix("data/rcicr_stimuli.Rdata",
baseimage = "base")The .rds is rebuilt automatically if you change the
source file (each cached file records the source’s size and modification
time, and is rebuilt when either changes). A once-per-session
cli line announces “cache built” or “cache reused”; silence
it with
options(rcisignal.silence_cache_messages = TRUE).
For the rcicr .Rdata reconstruction path, the upstream
rcicr package must be installed (it’s a Suggests).
Subsequent reads from the .rds cache do not need it.
validate_noise_matrix() runs basic sanity checks and
returns a diagnostic-style result rather than aborting:
validate_noise_matrix(nm,
expected_pixels = 256L * 256L,
expected_stimuli = 300L)4.4 The base image
The base face used at stimulus generation. Must be:
- Square (e.g. 256x256 or 512x512).
- Grayscale (single channel).
-
Pixel range
[0, 1](the conventionpng::readPNGandjpeg::readJPEGproduce). - Centred with eye/nose/mouth roughly at the geometry assumed by the default oval mask (eyes upper third, mouth lower third).
For a research-quality base, the webmorphR package by
DeBruine (2022) is the current best-in-class tool. The example below
uses R’s native pipe (|>, available since R 4.1) because
that is the idiom the webmorphR documentation uses; the rest of this
vignette sticks to base R for consistency.
library(webmorphR)
stim <- read_stim("path/to/raw_face_images/") |>
auto_delin() |> # automatic landmark delineation
align(procrustes = TRUE) |> # Procrustes alignment
crop(width = 0.85, height = 0.85) |> # tight crop
to_size(c(256, 256)) |> # rcicr-friendly size
greyscale() |>
avg() # morph into one average face
write_stim(stim, dir = "stimuli/", names = "base", format = "png")The output stimuli/base.png goes into
rcicr::generateStimuli2IFC(base_face_files = list(base = "stimuli/base.png")).
4.5 Face-region masks
rcisignal’s pixel-wise statistics aggregate or compare
across pixels, so the choice of which pixels enter the analysis
materially changes the reported number. A mask that includes hair and
background dilutes signal-localised effects roughly in proportion to the
area added.
Three ways to obtain a mask:
# 1. Parametric, no extra dependencies. Default oval geometry
# is a typical centred-face oval; tune via centre,
# half_width, half_height.
fm <- make_face_mask(c(256L, 256L), region = "full")
# Sub-regions for region-restricted analyses. Three of these
# (eyes, left_eye, right_eye) are axis-aligned rectangles
# tunable via the `region_bounds` argument; the rest are
# ellipses tunable via `centre`, `half_width`, `half_height`.
make_face_mask(c(256L, 256L), region = "eyes") # wide rectangle, both eyes
make_face_mask(c(256L, 256L), region = "left_eye") # rectangle, viewer's left eye
make_face_mask(c(256L, 256L), region = "right_eye") # rectangle, viewer's right eye
make_face_mask(c(256L, 256L), region = "mouth")
make_face_mask(c(256L, 256L), region = "nose")
make_face_mask(c(256L, 256L), region = "upper_face")
make_face_mask(c(256L, 256L), region = "lower_face")
# 2. From a hand-painted PNG / JPEG mask (e.g. from webmorphR or
# GIMP):
fm <- read_face_mask("masks/oval_256.png",
expected_dims = c(256L, 256L))
# 3. From a numeric matrix in code:
fm <- as.vector(custom_mask_matrix > 0.5)A mask can be supplied as either a logical vector of length
n_pixels (with pixels in the same order R uses when it
flattens a matrix into a vector, i.e. column by column) or as a logical
matrix with the image dimensions. Every mask argument in
the package accepts both forms.
plot_face_mask() renders any of those forms over the
base face, so you can verify alignment before passing the mask to a
metric:
plot_face_mask(fm, img_dims = c(256L, 256L),
base_image = "data/base.jpg",
main = "Full face oval (package default)")To overlay the mask directly on a specific base image (the workflow
you want when the question is “does this mask cover the right region of
this specific base image?”), use plot_mask_overlay():
# Either pass a prebuilt mask:
plot_mask_overlay(base_image = "data/base.jpg", mask = fm)
# Or use the `region =` shortcut to skip the make_face_mask()
# call. `region_bounds` is forwarded for rectangle-region tuning.
plot_mask_overlay(base_image = "data/base.jpg", region = "left_eye")Apply masks symmetrically. When a mask enters the
analysis, apply it to every term that goes into the statistic.
For infoval(), this means passing the mask to the function
so both the observed Frobenius norm and the reference distribution are
restricted to the same pixels. For rel_*() functions, pass
the mask via the mask argument; the package handles
symmetric application internally. Mixing a masked observed value with an
unmasked reference (or vice versa) yields a number that has no
defensible interpretation.
Visualising what a mask does to a base face
A mask is a logical vector that decides which pixels enter the analysis. Every pixel inside the mask contributes to the statistic; every pixel outside is ignored. Imposing a premade oval mask on the FMNES base face from the Karolinska Directed Emotional Faces stimuli (Lundqvist, Flykt, & Ohman, 1998), resized to 256 x 256, the visible difference is what is shown below.


Effect of a face-region mask on a base image. Left: raw FMNES base face
(Karolinska Directed Emotional Faces; Lundqvist et al. 1998). Right:
same face with a premade full-face oval mask applied; pixels outside the
mask are dimmed to light grey to make the analysed region explicit. The
reliability and discriminability metrics in this package will only see
the inside-mask pixels when a mask is supplied via the mask
argument.
When make_face_mask() is used to generate the mask
parametrically, eight region presets are available. Imposed on the same
base face (an artificial face generated with thispersondoesnotexist.com
so no consent or licensing concerns apply), they look as follows. Five
regions are ellipses (full, nose,
mouth, upper_face, lower_face);
the three eye regions (eyes, left_eye,
right_eye) are axis-aligned rectangles, tunable to a
specific base via the region_bounds argument (see the
tuning subsection below). All eight region geometries are this package’s
heuristics for a centred-portrait base; they are not taken from any
specific published paper. The convention of applying a full-face oval
before pixel-wise metrics follows prior practice in social-face RC
(e.g., Oliveira et al., 2019; Ratner et al., 2014; Schmitz, Rougier,
& Yzerbyt, 2024).








The eight built-in face-region masks rendered over the same
artificial-person base face (256 x 256). Each translucent red overlay
marks the pixels that pass through the mask; pixels outside are excluded
from the analysis. All eight region geometries are this package’s
heuristics for a centred-portrait base. The three rectangle eye regions
are independent of the full-oval geometry and tunable via
region_bounds.
The default geometry assumes the eyes sit roughly in the upper third
of the image and the mouth in the lower third (centred square base, face
filling most of the frame). Pass centre,
half_width, and half_height to
make_face_mask() if your base image has different
framing.
Tuning a sub-region for a non-default base face
The default sub-region geometry is calibrated for a centred, frontal
base face that fills most of the frame. The elliptical regions are
positioned relative to the full-face oval (centre,
half_width, half_height); the rectangle eye
regions are independent of the oval and tuned via their own
region_bounds. When the base image violates the
centred-portrait assumption, the parametric overlay drifts off the
intended feature and the metrics computed against it no longer mean what
their name implies.
There are two tuning routes, depending on the region’s shape:
-
Rectangle regions (
"eyes","left_eye","right_eye") take aregion_bounds = c(row_min, row_max, col_min, col_max)argument that sets the rectangle’s edges directly in 0-1 image fractions. Independent of the full-oval geometry; the rectangle for either eye can move without dragging the other along, which is the friction the previous two-ellipses-that-moved-together design ran into. -
Elliptical regions (
"full","nose","mouth","upper_face","lower_face") are positioned relative to the full-face oval and tuned via the globalcentre,half_width,half_height. For independent per-region adjustment of an ellipse (a common need with non-portrait or AI-generated bases), the exportedshift_mask()helper slides the mask by a number of pixels in any direction.
Rectangle regions: tune region_bounds
region_bounds accepts a length-4 numeric vector
c(row_min, row_max, col_min, col_max) in 0-1 image
fractions. Each pair must satisfy row_min < row_max and
col_min < col_max, and every entry must lie in
[0, 1]. The left and right eye rectangles are independent,
so each can be nudged separately to match a specific base.
# Tune just the viewer's left-eye rectangle on a base whose
# eye line sits a few percent below the heuristic default. The
# right-eye rectangle is unaffected.
left_eye_tuned <- make_face_mask(
c(256L, 256L), region = "left_eye",
region_bounds = c(0.40, 0.50, 0.24, 0.44)
)
# Verify the alignment visually before passing to a metric.
# plot_mask_overlay() also accepts a `region =` shortcut that
# builds the mask internally; pass region_bounds the same way.
plot_mask_overlay(base_image = "data/base.png",
region = "left_eye",
region_bounds = c(0.40, 0.50, 0.24, 0.44))If you measured the rectangle’s edges in pixels (by zooming into the
base image in your viewer or plot() window), use
region_bounds_from_pixels() to convert to the 0-1 fractions
region_bounds expects:
# "The viewer's left eye sits in rows 100-130, cols 60-115 on
# this 256-pixel base." Convert once, pass straight through.
bounds <- region_bounds_from_pixels(
row_min = 100, row_max = 130,
col_min = 60, col_max = 115,
img_dims = c(256L, 256L)
)
make_face_mask(c(256L, 256L), region = "left_eye",
region_bounds = bounds)Rendered over the artificial-person base used earlier, the default
left_eye rectangle and the tuned variant look as
follows:


Rectangle left_eye mask before and after tuning on a base
face whose eye line sits below the default. Left: default
region_bounds, sitting on the eyebrow. Right: nudged
downward by passing
region_bounds = c(0.40, 0.50, 0.24, 0.44). Because the
rectangle eye regions are independent of the full-oval geometry, the
right-eye rectangle would remain untouched.
Elliptical regions: global centre or per-region
shift
If every feature is offset in the same direction, pass the global
centre (and optionally half_width,
half_height) to make_face_mask():
# Whole-face shift: nose, mouth, and the full oval all move
# together. Rectangle eye regions are unaffected.
make_face_mask(c(256L, 256L), region = "mouth",
centre = c(0.55, 0.50)) # 5% downFor independent per-region tuning of an ellipse, the exported
shift_mask() helper slides the mask by a number of pixels
in any direction. Pixels shifted off the image are dropped, and the
newly exposed edge is filled with FALSE.
# Default mouth mask, reshaped from a flat logical vector
# (pixels in column-by-column order) back into a 256 x 256
# grid.
mouth_mask_default <- matrix(
make_face_mask(c(256L, 256L), region = "mouth"),
nrow = 256, ncol = 256
)
# Tune. On a 256-pixel image, 20 pixels is about 8 % of the
# height and 8 pixels is about 3 % of the width.
mouth_mask_v <- shift_mask(mouth_mask_default, down = 20)
mouth_mask_vh <- shift_mask(mouth_mask_default, down = 20, right = 8)shift_mask() accepts both down and
right and combines them in a single call, so vertical-only
and vertical-plus-horizontal tuning share the same idiom. It works on
either a column-major logical vector (pass img_dims) or a
logical matrix (returned in the same shape). Both infoval()
and the rel_*() family accept a logical matrix as the
mask argument, so the tuned grid can be passed in directly
without flattening.
Rendered over the same artif_base.png shown earlier
(where the mouth sits noticeably below the default), the default mask
and the two tuned variants look as follows:



Elliptical mouth-region mask before and after shift-tuning on a base
face whose mouth sits below the default. Left: default geometry. Middle:
shifted down by ~20 pixels (about 8 percent of image height). Right:
same vertical shift plus an ~8-pixel rightward shift. Each panel renders
one of the matrices produced by shift_mask() above. The
same recipe works for nose, upper_face,
lower_face, and the full oval; the three
rectangle eye regions use region_bounds instead.
Iterate with plot_mask_overlay() (overlay on the base
image) or plot_face_mask() until the overlay sits where you
want, then pass the tuned mask to infoval() or any
rel_*() function exactly as you would a parametric mask.
Useful shift magnitudes are typically a few pixels to a few dozen on a
256-pixel image; if you find yourself needing more than that, the whole
face is probably misaligned and centre should be retuned at
the global level via make_face_mask() instead.
5. Step 1: diagnose the inputs
Before computing CIs, run the diagnostic battery. The single entry
point is run_diagnostics(), which invokes every implemented
check whose required inputs are available and gathers the results into
one printable report.
5.1 A first run
The smallest meaningful call needs only the response data and the method:
report <- run_diagnostics(responses, method = "2ifc")
reportThe output looks like:
== Data-quality report (2ifc) ==
[PASS] Response coding
All 60,000 responses coded {-1, 1}.
[PASS] Trial counts
All 200 producers at 300 trials.
[PASS] Duplicates
No duplicate rows.
[PASS] Response bias
No constant responders, no |mean| > 0.6.
Summary: pass=4, warn=0, fail=0, skip=0
Skipped checks:
- check_rt (no col_rt)
- check_stimulus_alignment (no rdata or noise_matrix)
- check_version_compat (no rdata)
- compute_infoval_summary (no rdata + infoval_iter)
- check_response_inversion (no rdata + infoval_iter)
- check_rt_infoval_consistency (no rdata + infoval_iter + col_rt)The “Skipped checks” block is informational, not a failure: each listed check has prerequisites the call did not supply. The next section walks through how to unlock each.
5.2 The result object
run_diagnostics() returns an
rcisignal_diag_report with three fields:
-
$results: a named list ofrcisignal_diag_resultobjects, one per check that ran. -
$skipped_checks: character vector naming checks that were not run, each with the reason in parentheses. -
$method:"2ifc"or"briefrc".
Each rcisignal_diag_result has:
-
$status: one of"pass","warn","fail", or"skip". -
$label: short check name. -
$detail: character vector of explanation lines. -
$data: optional list of programmatic data (flagged participants, count tables, group-level statistics).
summary(report) returns a flat data frame with
check, status, label columns for
programmatic filtering. print() is the human-readable view
shown above.
5.3 The check_* family
Eight individual check functions cover the input-side battery. Each
takes responses plus its check-specific arguments and
returns an rcisignal_diag_result.
-
check_response_coding()verifies{-1, +1}coding. PASS for{-1, 1}; WARN with a recode formula for{0, 1}or{1, 2}; FAIL otherwise. The{0, 1}miscoding produced by experiment software that records “left” / “right” as 0 / 1 is a common silent failure in 2IFC. -
check_trial_counts(expected_n = ...)verifies that every producer has the expected number of trials.expected_ncan be a scalar or a named vector. PASS if all match; WARN at <= 10% off; FAIL above. -
check_duplicates()flags duplicate rows. PASS at 0; FAIL if >= 2 full duplicates and > 5% of rows; WARN otherwise. -
check_response_bias(bias_threshold = 0.6)flags constant responders (FAIL) and producers with|mean(response)| > bias_threshold(WARN; default 0.6 corresponds to roughly an 80/20 split). -
check_rt(col_rt = ...)scans response times for fast-clicking (default RT < 400 ms), implausibly slow trials, and low within-subject coefficient of variation. Defaults are conservative; tune them to your task. -
check_stimulus_alignment(rdata = ... | noise_matrix = ...)validates thatstimulusids fall inside the pool. FAIL on any out-of-range id; WARN if > 50% of the pool is unreferenced. -
check_version_compat(rdata = ...)(2IFC only) compares thegenerator_versionrecorded in the rdata to the installedrcicrversion. PASS if matching; WARN otherwise. The warning is informational (older datasets remain usable, and the flag simply prompts a spot-check). -
check_response_inversion(rdata = ..., infoval_iter = ...)detects whole-batch sign-flipped data by computing per-producer infoVal with the original responses and again with the negated responses. FAIL if >= 50% of producers are flagged as inverted; WARN if any are.
5.4 diagnose_infoval()
diagnose_infoval() is the recommended diagnostic for the
question “is my data informative at all?”. It runs a six-step
walk-through that catches every common low-infoVal cause:
- Compute observed Frobenius norm per producer (and group-mean).
- Compare against a reference distribution at each producer’s actual
trial count (closes the calibration gap in
rcicr::generateReferenceDistribution2IFC(), which keys on pool size). - Apply a face mask (default
"auto"= parametric full-face oval) and repeat. - Compare unmasked vs masked z to see whether masking lifts or depresses signal.
- Sanity-check with a synthetic random responder (should land at
|z| < 1). - Report whether the group-mean CI clears z = 1.96 even when per-producer medians do not.
iv <- diagnose_infoval(
responses,
method = "2ifc",
rdata = "rcic_stimuli.Rdata",
iter = 1000L,
face_mask = "auto",
seed = 1L
)
iv # PASS / WARN / FAIL with rich data attached to $dataThe status logic:
-
PASS: group-mean masked z >= 1.96 and
random-responder z is within
|z| < 1. Data is healthy. -
FAIL: random-responder
|z| > 2. Reference distribution is miscalibrated; almost always indicates a noise-matrix or pool-id mismatch. - WARN: anything in between. Usually means the per-producer signal is genuinely modest but the group CI is informative (typical pattern for trait inferences).
5.5 compute_infoval_summary()
A small convenience function around
rcicr::computeInfoVal2IFC() for the legacy 2IFC path. It
returns a per-participant z table plus a pass/warn summary, useful for
direct comparison with previously published rcicr numbers.
For the Brief-RC path or for the trial-count-matched reference, prefer
diagnose_infoval() or infoval() directly.
5.6 check_rt_infoval_consistency()
Cross-validates infoVal against RT quality by correlating per-producer infoVal with per-producer median RT. A strong negative correlation (correlation <= -0.30) suggests that fast clickers are also producing low-infoVal masks, indicating a population-level pattern rather than a single-producer fluke. WARN if the correlation passes the threshold; PASS otherwise.
5.7 Conditional checks and required arguments
When the call carries only response data, four checks run and six are skipped. Each skipped check requires a specific additional argument:
| Check | Required argument |
|---|---|
check_rt |
col_rt |
check_stimulus_alignment |
rdata (2IFC) or noise_matrix
(Brief-RC) |
check_version_compat |
rdata (2IFC only) |
compute_infoval_summary |
rdata + infoval_iter
|
check_response_inversion |
rdata + infoval_iter
|
check_rt_infoval_consistency |
rdata + infoval_iter +
col_rt
|
infoval_iter defaults to NULL because the
reference distribution simulation at 10,000 iterations takes minutes on
first call. Opt in explicitly when you are ready to wait.
report <- run_diagnostics(
responses,
method = "2ifc",
rdata = "rcic_stimuli.Rdata",
baseimage = "base",
col_rt = "rt",
expected_n = 300L,
infoval_iter = 1000L,
face_mask = "auto"
)With every input supplied, the “Skipped checks” block is empty.
6. Step 2: compute classification images
Once the diagnostics pass, compute CIs.
6.1 From raw responses
The 2IFC path delegates to rcicr::batchGenerateCI2IFC()
and returns a list with $signal_matrix (raw mask, ready for
rel_*), optionally $rendered_ci for
visualisation, plus metadata.
res <- ci_from_responses_2ifc(
responses,
rdata_path = "rcic_stimuli.Rdata",
baseimage = "base",
scaling = "none", # raw mask only; render later if needed
keep_rendered = FALSE
)
dim(res$signal_matrix) # n_pixels x n_participants
attr(res$signal_matrix, "source") # "raw"Behind the scenes the function takes care of the steps that are easy
to get wrong when calling rcicr directly: it loads the helper packages
rcicr expects (foreach, tibble,
dplyr), checks that responses are coded
{-1, +1}, runs single-threaded by default, and matches the
.Rdata filename in a case- insensitive way.
Brief-RC support is built into rcisignal directly (rcicr v1.0.1 does
not include Brief-RC). The implementation follows Schmitz’s
genMask() formula step for step, including the rule that
collapses repeated stimulus ids by averaging their responses:
res <- ci_from_responses_briefrc(
responses,
rdata_path = "rcic_stimuli.Rdata", # for the noise pool
base_image = "base.jpg", # path or numeric matrix in [0, 1]
method = "briefrc12"
)You can pass a pre-loaded noise_matrix instead of
rdata_path; useful when you have a non-rcicr-generated pool
(e.g. Schmitz’s OSF text matrix).
6.2 From pre-rendered CIs
When you already have one CI image per producer on disk (PNG or
JPEG), read_signal_matrix() reads them and subtracts the
base image in one call:
signal <- read_signal_matrix(
dir = "data/cis_condition_A/",
base_image_path = "data/base.jpg"
)
attr(signal, "source") # "rendered"read_cis() and extract_signal() are also
available on their own, for cases where you want to do something between
reading the PNGs and subtracting the base (e.g. masking, cropping, or
swapping the base image).
The first call to any Mode-1 reader emits the once-per-session
warning that PNG-derived signals are scaled. Silence with
options(rcisignal.silence_scaling_warning = TRUE) or pass
acknowledge_scaling = TRUE when calling.
6.3 CI scaling options
rcicr::batchGenerateCI2IFC() exposes a
scaling argument with five values:
-
"autoscale": stretches each producer’s mask to a fixed symmetric range. The rcicr default and the convention used in Schmitz et al. (2024) Experiment 2. -
"matched": stretches each mask to the base image’s range. Per-CI, so it breaks correlation-based metrics as well (a uniform scaling preserves Pearson, but a per-CI stretch does not). -
"independent": likeautoscalewith each CI’s stretch computed independently (no shared range across CIs). -
"constant": multiplies the mask by a fixed constant. -
"none": no scaling. Output isbase + raw_mask.
The shipped $signal_matrix is the raw unscaled mask
regardless of which scaling you pick; the
scaling argument only affects the optional
$rendered_ci field that keep_rendered = TRUE
returns.
Recommendation: feed the raw $signal_matrix to every
metric. For rcicr::computeInfoVal2IFC() the choice does not
matter (it reads $ci internally). For Brief-RC, treat any
non-none scaling as visualisation-only and never pass it to
rel_* or to hand-rolled infoVal.
7. Step 3: within-condition reliability
With the signal matrix in hand, the question is whether each condition’s group-level CI is stable: would you obtain the same group pattern from a different half of the producers? “Reliable” in the psychometric sense is shorthand for the producers’ CIs agree with each other enough that averaging them recovers the same pattern in repeated samples. Two complementary metrics address this question directly, alongside an influence-screening diagnostic that is sometimes confused with reliability.
The two reliability metrics:
-
rel_split_half()asks how well one random half of the producers reproduces the other half’s group CI. Repeating the split many times gives a sampling distribution for the agreement. -
rel_icc()asks how much of the pixel-by-producer signal variance is attributable to consistent producer-level patterns versus residual noise. It is the same intraclass correlation used in measurement theory and inter-rater reliability work.
The third metric (rel_loo()) is an influence screen: it
flags individual producers whose removal noticeably shifts the group CI,
useful for catching coding errors or outlier strategies but not itself a
reliability number.
A short note on what these metrics do not address. Reliability here is internal: would the same producers, if split differently, have produced the same CI? Whether the CI accurately captures the producer’s mental representation of the trait is a separate validity question, typically addressed by an external rater study, and sits outside the package.
7.1 rel_split_half()
Background. Split-half reliability is an old psychometric
trick (Spearman, 1910; Brown, 1910): if a measurement is internally
consistent, splitting it into two halves and correlating the halves
should give a high correlation. In the RC setting, the “halves” are two
random subsets of producers, and the quantities being correlated are the
pixels of the group-mean CI computed from each half. The catch is that
each half is built from N/2 producers rather than
N, so the half-half correlation underestimates the
reliability of the full N-producer CI. The Spearman-Brown correction
r_sb = (2 r_hh) / (1 + r_hh) projects the half-half
correlation up to the reliability the full sample would have if the
underlying signal really is shared. Repeating the split many times and
averaging both quantities reduces the dependence on any single random
partition.
In code, the function does exactly this:
Randomly partition the producers into two halves, compute the
group-level CI for each half (rowMeans()), correlate them,
and average across many permutations. The function reports both the mean
per-permutation r (r_hh) and the
Spearman-Brown projected full-sample reliability
(r_sb = (2 r_hh) / (1 + r_hh)). The headline number is
typically r_sb.
sh <- rel_split_half(signal_matrix,
n_permutations = 2000L,
seed = 1L)
sh
plot(sh)Permutation is over producers (not pixels) so that each producer’s
spatial structure is preserved. For odd N, one randomly-chosen producer
is dropped per permutation (re-drawn each iteration) so both halves
contain floor(N/2) producers.
The null argument adds an empirical chance baseline:
-
null = "permutation": per iteration, generates fresh Gaussian noise per producer (no shared spatial structure), then recomputesr_hh. Centred at 0 and useful as a worst-case floor. -
null = "random_responders": simulatesncol(signal_matrix)random responders using the samegenMask()machinery asinfoval()’s reference. This baseline preserves the pixel correlation structure of real noise patterns and tracks the empirical chance baseline of an actual RC experiment more closely. Requiresnoise_matrix.
sh <- rel_split_half(signal_matrix,
null = "random_responders",
noise_matrix = nm,
n_permutations = 2000L,
seed = 1L)
sh$r_hh # observed
sh$r_hh_null_p95 # 95th percentile of the null
sh$r_hh_excess # observed - null median
sh$r_sb_excess # same, projected via Spearman-BrownReport $r_sb as the headline; $r_sb_excess
as the above-chance increment when a null is requested.
$ci_95 / $ci_95_sb are percentile 95% CIs on
the observed distribution.
rel_split_half_null() exposes the same null-distribution
simulation as a standalone function, useful when you want to precompute
a null and reuse it across conditions with the same producer count.
7.2 rel_icc()
Background. The intraclass correlation coefficient (ICC) is a family of statistics for asking how much of the variability in repeated measurements is attributable to differences between the objects of measurement (here, producers) versus residual noise (McGraw & Wong, 1996; Koo & Li, 2016). Every producer has one CI vector with one entry per pixel, and the ICC asks how consistently the producers agree on that pixel-by-pixel pattern. A high ICC means producers’ CIs are similar to each other relative to noise; a low ICC means they are not.
The “(3,*)” label is McGraw & Wong’s notation for a two-way
mixed-effects model in which the column factor (pixels in our case) is
fixed and the row factor (producers) is random. The fixed-pixels choice
reflects the reality of an RC experiment: the image grid is not a random
sample from a population of pixels; it is the same set of pixels across
all producers. The “3,1 vs 3,k” distinction is whether you want the
reliability of a single producer’s CI (3,1) or of the
group-averaged CI across k producers
(3,k).
rel_icc() returns both, computed from a two-way mixed
model with pixels fixed and producers random:
- ICC(3,1) answers “how informative is one producer’s CI as a noisy estimate of the group pattern?”.
-
ICC(3,k) answers “how stable is the group-mean CI
across
kproducers?”. Usually the headline.
ic <- rel_icc(signal_matrix)
ic # prints ICC(3,1), ICC(3,k), MS rows / cols / errorThe function computes both quantities directly from ANOVA mean
squares, which scales to large image grids that would otherwise run out
of memory. Results agree with psych::ICC() on smaller
matrices where both can be run.
ICC(3,) is appropriate when pixels are fixed. ICC(2,)
(two-way random) treats pixels as a random sample from a pixel
population, which the image grid is not, even when ICC(2,) and
ICC(3,) give similar numbers at high pixel counts. Use
variants = c("3_1", "3_k", "2_1", "2_k") to report ICC(2,*)
side-by-side when comparability with reports that use the two-way-random
model is needed.
ICC is variance-based, so it errors on a "rendered"
source matrix unless acknowledge_scaling = TRUE is passed.
Rendered scaling corrupts ICC values in non-recoverable ways, so the
default behaviour is conservative.
A once-per-session warning fires when
n_targets > 50,000 and ICC(3,k) is requested, flagging
that ICC(3,k) tends toward 1 at large image sizes (it is not
resolution-comparable). Report ICC(3,1) as the primary statistic for
cross-resolution comparisons.
Empirically, ICC(3,k) and the group-mean infoVal z (see §9) track each other very closely on real data: both quantify how aligned the producers are on the pixel-level signal, ICC(3,k) as a variance ratio and group-mean z as the magnitude of the surviving group-mean signal against a matched reference. §12.6 shows the empirical relationship across ten trait conditions on the Oliveira et al. (2019) data (Pearson r ≈ 0.97).
7.3 rel_loo()
Background. Leave-one-out (LOO) is a jackknife-style
influence diagnostic (Tukey, 1958; Cook, 1977): for each producer in
turn, recompute the group CI with that producer removed and correlate
the result with the full-sample group CI. Producers whose removal
changes the group CI more than the others stand out as high-influence
cases worth a closer look. This is not a reliability
metric. The with-producer and without-producer group CIs share
(N-1)/N of their data, so LOO correlations are near 1 by
construction even on noisy data; the diagnostic information lives in the
relative ordering of producers, which the function summarises
as a robust z-score.
For each producer i, this function computes the Pearson
correlation between the full-sample group CI and the group CI recomputed
without producer i. Producers whose r_loo sits
well below the others are candidates for inspection.
lo <- rel_loo(signal_matrix)
lo # raw cors + z-scores + flag column
rel_loo_z(lo) # tidy data frame, sorted by z_score
plot(lo)rel_loo() is an influence-screening diagnostic, distinct
from the reliability metrics in §7.1 and §7.2. Because the full-sample
mean and the leave-one-out mean share (N-1)/N of their
data, r_loo values are near 1 by construction even on noisy
data (typically in the [0.95, 0.999] range at N = 30). The
relative ordering across producers carries the diagnostic information,
so the function reports $z_scores as the recommended
quantity.
Two flagging rules are available: "mad" (default) and
"sd" (kept for compatibility with earlier versions; will be
removed in v0.2.0). MAD is robust to the influential producers the test
is meant to flag; SD’s mean and standard deviation are themselves pulled
by the outlier. Default flag_threshold = 2.5 so that a
30-producer dataset flags ~0.3 producers by chance.
A flag prompts inspection rather than exclusion. Investigate first
(response coding, fatigue, atypical strategy) and cross-check with
run_diagnostics() to rule out coding errors before
excluding any producer.
7.4 run_reliability()
Top-level convenience function that runs
rel_split_half() and rel_icc() on a single
signal matrix and bundles both results into one
rcisignal_rel_report:
rep <- run_reliability(signal_matrix,
n_permutations = 2000L,
seed = 1L)
rep
plot(rep)rep$results$split_half and rep$results$icc
are the standalone result objects. run_reliability()
deliberately omits rel_loo(), since LOO is an
influence-screening diagnostic and bundling it into a reliability report
invites misreading r_loo’s near-1 values as
reliability.
8. Step 4: between-condition discriminability
When the design has two or more conditions, the question becomes whether their group CIs are distinguishable, both in overall magnitude and in spatial location.
The hard part of doing this honestly is the multiple comparisons
problem. A typical RC image has tens of thousands of pixels (a 256
x 256 grid has 65,536). If you compute one t-test per pixel and
threshold each at the conventional p < 0.05, you should
expect about 5% of pixels to come out “significant” by chance even when
the two conditions are identical, which is over 3,000 false-positive
pixels per image. The naive Bonferroni correction (divide alpha by the
number of tests) overcorrects in the opposite direction because pixels
are spatially correlated; CI signal lives in contiguous regions, not
independent dots.
The solution adopted here is borrowed from the neuroimaging literature: build a single test statistic that summarises the spatial extent of the effect (a cluster), and calibrate it against a null distribution generated by random label permutation. This controls the family-wise error rate (FWER) in the strong sense, that is, the probability of one or more false positives anywhere in the image is held below the chosen alpha (Nichols & Holmes, 2002; Maris & Oostenveld, 2007). Two test statistics are offered:
- A classical cluster-mass statistic with a user-set cluster- forming
threshold (
rel_cluster_test(method = "threshold")). - Threshold-free cluster enhancement, TFCE
(
rel_cluster_test(method = "tfce"); Smith & Nichols, 2009), which sidesteps the threshold choice by integrating the cluster contribution to each pixel across many thresholds.
A complementary question is the overall magnitude of the
difference, independent of where in the image it sits.
rel_dissimilarity() addresses that with a single distance
number plus a bootstrap confidence interval.
8.1 pixel_t_test()
Background. The pixel-wise t-test is the building block of everything in this section: at each pixel, compare the two conditions’ producer-level signal values with a Welch’s t (or a paired t when producers are matched). The output is one t-value per pixel, an image-shaped statistical map of where the conditions differ.
This map is informative, but it is not a valid statistical test on
its own. With tens of thousands of pixels and no multiplicity
correction, raw per-pixel p-values overstate the strength of evidence
(see the §8 intro above). For that reason pixel_t_test()
returns the t-vector and stops there; turning it into inference is the
job of rel_cluster_test() (§8.2), which feeds these
per-pixel t-values into a permutation-based FWER procedure.
In code:
Vectorised Welch’s t (independent groups) or paired t (matched producers) per pixel:
t_vec <- pixel_t_test(signal_a, signal_b) # n_pixels long
t_vec_paired <- pixel_t_test(signal_a, signal_b,
paired = TRUE)Returns a numeric vector of t-values, length n_pixels
(or sum(mask) if a mask is supplied). The function serves
as an intermediate building block for rel_cluster_test()
and is not intended as a standalone inferential test (no FWER control at
the per-pixel level). For paired mode, the two matrices must have
identical ncol and matching column names.
8.2 rel_cluster_test()
Background. The cluster-based permutation test (Maris & Oostenveld, 2007) treats the spatial coherence of CI signal as a feature, not a nuisance. The procedure is in three conceptual steps:
- Compute the per-pixel t-statistic (as in §8.1), and threshold its
absolute value at some cutoff (e.g.,
|t| > 2). Each pixel is either supra-threshold or not. - Group the supra-threshold pixels into spatially contiguous
clusters using a neighbour rule. The default in
rcisignalis 4-connectivity: a pixel’s neighbours are the four pixels sharing an edge with it (up, down, left, right). 8-connectivity (which also counts diagonal neighbours) tends to merge things that only touch at a corner, producing larger and fewer clusters; 4-connectivity is the conservative default and is the one used here. - Score each cluster by its mass: the sum of the t-values inside the cluster. A small cluster of very high t-values can have the same mass as a large cluster of moderate t-values, and the mass statistic treats them as comparably strong evidence.
The hard part is calibration: how big a cluster mass would you expect
under the null (the conditions are identical)? The permutation
answer is to randomly relabel which producer is in which condition
(preserving the per-condition N), recompute the per-pixel t
and the maximum cluster mass, and repeat thousands of times. The
resulting distribution of maximum cluster masses is the empirical null,
calibrated to your data without distributional assumptions. A
real cluster’s p-value is the fraction of null max-masses that exceed
it. Because every candidate cluster is tested against the
maximum under the null, this controls the family-wise error
rate in the strong sense (Nichols & Holmes, 2002).
A note of self-criticism: the cluster-forming threshold is the one knob without a fully principled choice. Lower thresholds favour broad and diffuse effects; higher thresholds favour focal and intense ones (Smith & Nichols, 2009). When you do not have prior intuition about the spatial scale of your effect, TFCE (below) avoids having to choose.
In code, rel_cluster_test() exposes both methods.
method = "threshold" (default; Maris
& Oostenveld 2007): threshold
|t| > cluster_threshold (default 2.0), label connected
components with 4-connectivity (the conservative choice over
8-connectivity), and use cluster mass (sum of t-values within the
cluster, not pixel count) as the test statistic. The null is built by
stratified label permutation: every permutation preserves
(N_a, N_b) exactly, the pixel-wise t is recomputed on
shuffled labels, and the maximum positive and maximum negative cluster
masses are recorded. A cluster’s p-value is the fraction of null masses
(matching sign) that exceed the observed.
ct <- rel_cluster_test(
signal_a, signal_b,
img_dims = c(256L, 256L),
cluster_threshold = 2.0,
n_permutations = 2000L,
alpha = 0.05,
seed = 1L
)
ct
plot(ct)The result carries $clusters (a data frame with
cluster_id, direction, mass,
size, p_value, significant),
$null_distribution (the $pos and
$neg per-permutation max masses), and integer label
matrices $pos_labels / $neg_labels for
plotting. Maximum-statistic permutation provides FWER control in the
strong sense (Nichols & Holmes 2002).
method = "tfce" (Smith & Nichols
2009): threshold-free cluster enhancement. Instead of picking one
cluster-forming threshold, TFCE walks across many possible thresholds
and adds up, at each pixel, how much spatial support the surrounding
cluster has at each threshold height. Pixels embedded in
strongly-supported clusters end up with high TFCE scores; isolated
pixels with no neighbour support end up with low scores. Per-pixel TFCE
value is the integral over thresholds of size^E x h^H x dh;
positive and negative tails are enhanced separately and recombined with
sign preserved. No free threshold parameter to choose. Per-pixel p-value
=
(sum(null_max_abs_tfce >= |observed_tfce|) + 1) / (n_perm + 1).
ct_tfce <- rel_cluster_test(
signal_a, signal_b,
img_dims = c(256L, 256L),
method = "tfce",
tfce_H = 2.0,
tfce_E = 0.5,
seed = 1L
)Defaults match Smith & Nichols (H = 2.0,
E = 0.5, n_steps = 100). TFCE result carries
$tfce_map, $tfce_pmap,
$tfce_significant_mask instead of $clusters.
Print and plot methods branch on $method.
For a paired design, pass
paired = TRUE; the per-pixel statistic becomes paired t and
the null is built by random sign-flip on per-producer differences (exact
under exchangeability of pair sign).
8.3 rel_dissimilarity()
Background. The Euclidean distance between two CIs is the
square root of the summed squared pixel differences (equivalently, the
Frobenius norm of the difference matrix). It is a single- number summary
of how far apart two group CIs sit in pixel space. It ignores
where the difference lives and reports only its overall
magnitude. The advantage is simplicity: one number per contrast,
comparable across studies, with a percentile bootstrap CI (resample
producers with replacement, recompute the distance, repeat) for
uncertainty. The trade-off is loss of spatial detail, which is exactly
what rel_cluster_test() (§8.2) is for. Reported together,
the two metrics complement each other: the cluster test localises the
difference, the dissimilarity quantifies its overall size.
A self-critical note: Euclidean distance scales with image size and
with whatever absolute units the CI carries. The
$euclidean_normalised field divides by
sqrt(n_pixels) to make distances comparable across
resolutions, but cross-study comparison still requires care about
scaling conventions and the underlying base image.
In code:
Pair the spatial cluster test with a single overall magnitude summary: Euclidean distance between the two group-mean CIs, with percentile bootstrap CIs.
dr <- rel_dissimilarity(
signal_a, signal_b,
n_boot = 2000L,
ci_level = 0.95,
seed = 1L
)
dr
plot(dr)$euclidean is the raw distance;
$euclidean_normalised is
$euclidean / sqrt(n_pixels), useful for cross-resolution
comparisons. $boot_dist is the full bootstrap distribution;
$ci_dist and $boot_se_dist are the standard
summaries.
The Pearson correlation fields ($correlation,
$boot_cor, $ci_cor, $boot_se_cor)
are kept for compatibility with earlier versions but are no longer
recommended. Two base-subtracted CIs share image-domain spatial
structure (face shape, oval signal support) that pushes their
correlation above zero even when the underlying mental representations
are unrelated; absolute correlation values do not cleanly mean “these
conditions are similar”.
A null = "permutation" argument adds a chance baseline
for the Euclidean distance: stratified condition-label permutation
(between-subjects) or sign-flip on per-producer differences (paired).
When set, the result includes $d_null_p95,
$d_z (z-equivalent effect size), and
$d_ratio.
8.4 run_discriminability()
Orchestrator that runs rel_cluster_test() and
rel_dissimilarity() on a pair of signal matrices:
rep <- run_discriminability(signal_a, signal_b,
img_dims = c(256L, 256L),
cluster_threshold = 2.0,
seed = 1L)
rep
plot(rep) # cluster t-map + bootstrap dissimilarity, side by sideplot(rep) is the one-call view of the whole report. The
children rep$results$cluster_test and
rep$results$dissimilarity are the standalone results,
useful when you want a single panel with a custom title (for example to
compare conditions named on a poster or a figure caption):
8.5 run_discriminability_pairwise()
Generalises run_discriminability() to all K-choose-2
pairs of K conditions, with a family-wise correction across pairs (Holm
by default, Bonferroni or none also available):
rep <- run_discriminability_pairwise(
signal_matrices = list(
Trust = sm_trust,
Dominant = sm_dominant,
Friendly = sm_friendly
),
fwer = "holm",
seed = 1L
)
rep$pairs # one row per pair: cond_a, cond_b, p_min, p_adj_pair
plot(rep) # cluster t-map per pair, laid out in a gridplot(rep) lays out one cluster t-map per pair in a
square-ish grid. A warning fires above max_pairs = 12
because at that density the panels become illegible; subset
rep$results and plot a slice, or call
plot(rep, max_pairs = Inf) to silence the warning. To
inspect a single pair on its own, or to override the title:
plot(rep$results[["Trust_vs_Dominant"]]$cluster_test,
main = "Trust vs Dominant")
plot(rep$results[["Trust_vs_Dominant"]]$dissimilarity,
main = "Trust vs Dominant - bootstrap")For a shared-axis comparison of bootstrap distances across pairs,
pass each pair’s dissimilarity child to
plot_dissimilarity_grid().
The per-pair statistic carried into the across-pairs adjustment is
the minimum cluster-level p-value within each pair;
within-pair cluster p-values are not re-adjusted (they retain the
max-statistic FWER control from the underlying
rel_cluster_test()). A pair with no clusters contributes
p_min = 1.0.
9. Step 5: per-producer informational value
Background. Per-producer infoVal (Brinkman et al., 2019)
asks a single question: is this producer’s CI larger in magnitude than
what a random responder would produce on the same task? The metric is a
z-score. The numerator is the Frobenius norm of the CI
mask (the square root of the summed squared pixel values, a scalar that
summarises overall mask “size”). The reference distribution comes from
simulating many random (stimulus, ±1) response sequences
through the same CI formula, computing the Frobenius norm of each
simulated mask, and recording the mean and standard deviation of those
norms. A producer’s infoVal of, say, 2.5 means their CI is 2.5 standard
deviations larger than the random-responder mean; the conventional
threshold for “more than chance” is z >= 1.96, the
standard one-sided 0.05 cutoff.
What infoVal does not tell you is whether the CI points at the right pattern. The Frobenius norm is a magnitude statistic only. A producer who clicked consistently but in a way that encodes an unrelated visual pattern (for example, their CI looks like an upper-left versus lower-right contrast rather than a trait-relevant face feature) can still score high on infoVal. Pair infoVal with the reliability metrics (§7) and the discriminability metrics (§8) when you want a fuller picture.
A note on group-mean infoVal. The package also offers a
group-mean version (group_mean_z(), called inside
diagnose_infoval() as a supplementary statistic). It
applies the same Frobenius-norm logic to the group-averaged CI, using a
matched-N reference distribution so the calibration is fair (see §15 for
what goes wrong when that reference is mismatched). Brinkman et
al. (2019) recommend reporting the distribution of per-producer
infoVals as the primary group-level summary, not a single group z. The
group-mean version is offered here as a supplementary headline number
and is not independently validated for social-face RC; treat it as
exploratory (§1.2).
In code:
infoval() reports a per-producer Frobenius-norm z-score
against a reference distribution matched to that producer’s actual trial
count:
iv <- infoval(
signal_matrix,
noise_matrix,
trial_counts, # named integer vector matching colnames
iter = 10000L,
mask = make_face_mask(c(256L, 256L)),
seed = 1L,
cache_path = "data/infoval_cache.rds"
)
iv$infoval # named numeric: per-producer z-score
plot(iv)The function unifies 2IFC and Brief-RC infoVal under one
implementation. The only difference between paradigms is what you pass
as noise_matrix. The reference distribution is built per
unique trial count by simulating random (stim, ±1) pairs
through Schmitz’s genMask() formula and computing Frobenius
norms of the resulting masks. Producers sharing a trial count share a
reference, so the simulation cost is paid once per distinct trial count
rather than once per producer.
Trial-count matching closes a calibration gap. The
original rcicr::generateReferenceDistribution2IFC() keys
its reference on the full pool size. For 2IFC this is appropriate (every
producer typically responds to every pool item). For Brief-RC, where the
recorded number of mask contributions equals n_trials and
is typically smaller than n_pool, a pool-size reference
biases infoVal downward. infoval() uses each producer’s
actual recorded trial count.
Interpreting infoVal. The Frobenius norm is a magnitude statistic, answering “is this mask larger than chance?” rather than “is it pointing at the right pattern?”. Two consequences follow.
- Cross-paradigm comparisons require care. Brief-RC and 2IFC are placed on the same z-scale, but the cognitive processes generating the masks differ. A producer who benefits from Brief-RC’s richer per-trial context might produce a more accurately localised yet smaller-magnitude mask, which the metric will not reward.
- Stability and discriminability are addressed by other metrics.
rel_split_half()andrel_icc()quantify whether the signal is stable;rel_cluster_test()andrel_dissimilarity()quantify whether conditions are separable. Pairinfoval()with these to triangulate.
If most per-producer z-scores in your data come out near zero or negative, that is not unusual; see Appendix §15 for a five-reason walkthrough and a diagnostic recipe before drawing conclusions about producers’ engagement.
The with_replacement argument controls how stimulus ids
are drawn when simulating a random producer. "auto"
(default) matches the standard Brief-RC convention (without replacement
when the producer’s trial count fits in the pool). Set explicitly only
when your design departs from this convention.
The cache_path mechanism stores the reference norms in
an .rds file, indexed by the simulation settings
(iter, pool size, mask, and with_replacement).
If you call infoval() again with the same settings, it
loads the cached reference; otherwise it recomputes.
10. Step 6: agreement maps and paper figures
Three plot helpers turn results into publication-grade figures.
10.1 agreement_map_test()
Within a single condition, tests at each pixel whether the
producer-level signal differs from zero (one-sample t). The null is
built by random sign-flip per producer (exact under the assumption that,
under H0, the producer’s signal is symmetric around zero). Family-wise
error is controlled by the maximum |t| statistic across
pixels.
am <- agreement_map_test(signal_matrix,
n_permutations = 5000L,
alpha = 0.05,
mask = make_face_mask(c(256L, 256L)),
seed = 1L)
am$significant_mask # logical vector: which pixels survive FWER10.2 plot_agreement_map()
Renders the per-pixel one-sample t-map as a colour image, with optional thresholding:
plot_agreement_map(signal_matrix,
img_dims = c(256L, 256L),
threshold = 2.0,
palette = "diverging")10.3 plot_ci_overlay()
The headline figure for most papers. Renders the group-mean CI as a
translucent layer over the base face, optionally restricted to the
significant-pixel mask returned by
agreement_map_test():
plot_ci_overlay(
signal_matrix,
base_image = "data/base.jpg",
mask = am$significant_mask,
alpha_max = 0.7
)10.4 plot_dissimilarity_grid()
Lays out multiple rel_dissimilarity() results
side-by-side as labelled CI bars. Useful for paper figures showing
whether two contrasts have overlapping CIs without forcing the reader to
read four numbers from a table:
d_AB <- rel_dissimilarity(sm_a, sm_b, seed = 1L)
d_AC <- rel_dissimilarity(sm_a, sm_c, seed = 1L)
plot_dissimilarity_grid(
"Trust vs Dominant" = d_AB,
"Trust vs Competent" = d_AC,
metric = "euclidean_normalised"
)11. Region-restricted analyses
Every rel_*() and infoval() accepts a
mask argument. When you supply one:
-
rel_*()row-subsets the signal matrix to the masked pixels before computing the statistic. The reportedn_pixelsreflects the subsetted count. -
rel_cluster_test()uses a zero-out pattern instead, setting per-pixel t to 0 outside the mask. This preserves the 2D image structure required for 4-connectivity and TFCE. -
infoval()applies the mask symmetrically to both the observed Frobenius norm and the reference distribution. -
agreement_map_test()row-subsets and embeds the result back into a full-image vector (NA outside the mask).
The same mask object should pass through all metrics in a single analysis. Mixing masked observed values with an unmasked reference yields a number with no defensible interpretation. To compare across regions, run the metric once per mask:
for (region in c("eyes", "nose", "mouth",
"upper_face", "lower_face")) {
m <- make_face_mask(c(256L, 256L), region = region)
cat(region, ": ICC(3,1) =",
rel_icc(signal_matrix, mask = m)$icc_3_1, "\n")
}12. Worked example: Oliveira et al. (2019), Study 1
This section runs the package end-to-end on a published 2IFC dataset. The original paper reports the per-trait classification images and judge ratings. The reliability, discriminability, infoVal, and region-restricted analyses below are new and extend that work; the package post-dates the paper. Numbers and figures below come from running the package on the open data.
Oliveira, M., Garcia-Marques, T., Dotsch, R., & Garcia-Marques, L. (2019). Dominance and competence face to face: Dissociations obtained with a reverse correlation approach. European Journal of Social Psychology. https://doi.org/10.1002/ejsp.2569. Open data: https://doi.org/10.17605/osf.io/hr5pd.
In Study 1, 200 participants completed a 2IFC reverse-correlation task with 300 trials each on a 256 x 256 grayscale male base face, across 10 trait conditions in a between-subjects design (20 producers per trait): Dominant, Submissive, Trust, Untrust, Friendly, Unfriendly, Intelligent, Unintelligent, Competent, Incompetent.
The R code chunks below are shown for reading and adaptation; they
are marked eval = FALSE to keep the vignette quick to
render. The numbers and figures shown alongside each chunk were
precomputed by the data-raw/oliveira_2019/precompute*.R
scripts on the open OSF data and are loaded into the vignette via
readRDS() and knitr::include_graphics().
Re-run the precompute scripts to refresh after package changes.
12.1 Loading the data
The original CSV is semicolon-separated. We read it with
read.csv2(), then rename subject to the column
name the package expects, store the ids as text (so they are not treated
as numeric), and lower-case the trait labels for consistency:
library(rcisignal)
library(dplyr)
raw <- read.csv2("study1data.csv", stringsAsFactors = FALSE)
raw$participant_id <- as.character(raw$subject)
raw$trait <- tolower(raw$trait)
raw <- raw[, c("participant_id", "trial",
"stimulus", "response", "trait")]
head(raw)
#> participant_id trial stimulus response trait
#> 1 8001 1 152 1 dominant
#> 2 8001 2 284 -1 dominant
#> 3 8001 3 176 1 dominant
#> ...12.2 Modernising the legacy rcicr 0.3.0 rdata
The 2015 rdata stores its noise basis under s$sinusoids
and s$sinIdx, while current rcicr expects the
p$patches and p$patchIdx schema introduced in
v1.0.x. Patch the legacy file without re-running stimulus
generation:
load("rcic_seed_1_time_fev_05_2015_03_17.Rdata") # legacy file
p <- list(
patches = s$sinusoids,
patchIdx = s$sinIdx,
noise_type = "sinusoid"
)
save(list = ls(), file = "stimuli_modernised.RData")The new stimuli_modernised.RData is the file the package
will read.
12.3 Diagnostics
The diagnostic battery runs in one call:
report <- run_diagnostics(
raw[, c("participant_id", "stimulus", "response")],
method = "2ifc",
rdata = "stimuli_modernised.RData",
expected_n = 300L
)
print(report)Loaded from cache, the report’s summary on this dataset:
| check | status | label |
|---|---|---|
| response_coding | pass | Response coding |
| trial_counts | pass | Trial counts |
| duplicates | pass | Duplicates |
| response_bias | pass | Response bias |
| stimulus_alignment | pass | Stimulus alignment |
| version_compat | warn | rcicr version compatibility |
The version warning (when present) is informational and expected on this dataset because the experiment was run with rcicr 0.3.x in 2015. The basic mechanics are clean.
Some research designs cross multiple conditions, in which case
running check_response_bias() separately per condition is
useful (a producer who looks balanced overall may still be heavily
biased in one trait):
trait_bias <- list()
for (tr in sort(unique(raw$trait))) {
sub <- subset(raw, trait == tr,
c("participant_id", "stimulus", "response"))
trait_bias[[tr]] <- check_response_bias(sub, method = "2ifc")
}
trait_bias[["competent"]]For this dataset, all ten trait conditions return PASS.
12.4 Per-trait infoVal
infoval() reports a per-producer Frobenius-norm z-score
against a trial-count-matched reference. Running it on each of the ten
trait conditions, masked with the package’s default full-face oval,
gives the table below (precomputed):
| Trait | Median producer z | n above 1.96 (of 20) | Group-mean z |
|---|---|---|---|
| competent | +0.70 | 3 | +3.03 |
| dominant | +0.89 | 6 | +7.09 |
| friendly | +0.97 | 5 | +18.50 |
| incompetent | +0.59 | 2 | +2.13 |
| intelligent | +0.67 | 3 | +8.23 |
| submissive | +0.37 | 5 | +3.78 |
| trust | +0.50 | 3 | +11.95 |
| unfriendly | +0.85 | 5 | +15.73 |
| unintelligent | +0.63 | 3 | +2.03 |
| untrust | +0.84 | 7 | +9.58 |
Group-mean z is the headline. Per-producer median z sits well below 1.96 across all ten conditions, while the group-mean z typically clears it. This pattern is structural rather than a data problem: per-producer Frobenius norms aggregate over the whole image and dilute localised signal, so individual z values are systematically smaller than the group-mean equivalent on the same data. Brinkman et al. (2019) report the same general pattern for trait inferences.
12.5 Building one signal matrix, step by step
A signal matrix has one row per pixel and one column per producer. Each column is that producer’s mean noise pattern, sign-weighted by their responses across the trials they saw. The fastest way to understand the mask formula is to build one condition’s signal matrix by hand. We will do it for the Trust condition.
Step 1. Read the noise matrix once. Each column is the noise pattern shown on one trial out of the 300-stimulus pool.
noise_matrix <- read_noise_matrix("stimuli_modernised.RData",
baseimage = "male")
dim(noise_matrix)
#> 65536 x 300 # n_pixels x pool_sizeStep 2. Subset the response data to the Trust condition, sort by producer and trial, and read out the producer ids.
trust_trials <- raw[raw$trait == "trust", ]
trust_trials <- trust_trials[order(trust_trials$participant_id,
trust_trials$trial), ]
trust_ids <- unique(trust_trials$participant_id)
length(trust_ids)
#> 20Step 3. Compute one producer’s mask. Take the noise
patterns that producer saw (noise_matrix[, p$stimulus]),
multiply each by their response (+1 or -1) and
divide by the trial count. The result is a numeric vector of length
65,536 (the pixels in column-major order, i.e. column by column).
p1 <- trust_trials[trust_trials$participant_id == trust_ids[1], ]
# One column of `noise_matrix` per trial that producer saw,
# in trial order:
selected_noise <- noise_matrix[, p1$stimulus]
# Sum each pixel across trials weighted by the +/- 1 response,
# then divide by the trial count. The result is the producer's
# mean noise pattern, sign-weighted by their responses.
mask_1 <- (selected_noise %*% p1$response) / nrow(p1)
length(mask_1)
#> 65536Step 4. Repeat across all 20 Trust producers and
stack the results column-wise. Tag the matrix with img_dims
so plot helpers know it is 256 x 256, and with
source = "raw" so variance-based metrics will accept it
without complaint.
# Empty 65,536 x 20 matrix; one column per producer.
sm_trust <- matrix(NA_real_, nrow = nrow(noise_matrix),
ncol = length(trust_ids),
dimnames = list(NULL, trust_ids))
# Fill in one column per producer using the same recipe as Step 3.
for (i in seq_along(trust_ids)) {
p_i <- trust_trials[trust_trials$participant_id == trust_ids[i], ]
selected_noise <- noise_matrix[, p_i$stimulus]
sm_trust[, i] <- (selected_noise %*% p_i$response) / nrow(p_i)
}
# Tag image dimensions (so plot helpers know it is 256 x 256) and
# mark the matrix as a raw mask (so variance-based metrics accept it).
attr(sm_trust, "img_dims") <- c(256L, 256L)
attr(sm_trust, "source") <- "raw"
dim(sm_trust)
#> 65536 x 20That is the full recipe. The other nine conditions use the same
recipe with a different trait label. To produce all ten in
one shot, wrap the four steps in a function and apply it across all the
trait labels:
build_signal_matrix <- function(raw, label, noise_matrix) {
trials <- raw[raw$trait == label, ]
trials <- trials[order(trials$participant_id, trials$trial), ]
ids <- unique(trials$participant_id)
m <- matrix(NA_real_, nrow = nrow(noise_matrix),
ncol = length(ids),
dimnames = list(NULL, ids))
for (i in seq_along(ids)) {
p_i <- trials[trials$participant_id == ids[i], ]
selected_noise <- noise_matrix[, p_i$stimulus]
m[, i] <- (selected_noise %*% p_i$response) / nrow(p_i)
}
attr(m, "img_dims") <- c(256L, 256L)
attr(m, "source") <- "raw"
m
}
traits <- sort(unique(raw$trait))
sm <- lapply(traits, function(tr)
build_signal_matrix(raw, tr, noise_matrix))
names(sm) <- traits
sm_trust <- sm[["trust"]]
sm_dominant <- sm[["dominant"]]
sm_competent <- sm[["competent"]]
sm_friendly <- sm[["friendly"]]In a real pipeline, ci_from_responses_2ifc() performs
the same work and additionally handles the rcicr
integration. Doing it by hand once makes the mask formula concrete; you
can switch to ci_from_responses_2ifc() afterwards.
12.6 Within-condition reliability per trait
run_reliability() returns split-half (with
Spearman-Brown projection) and ICC(3,*) on a single signal matrix. Run
it on each per-trait signal matrix:
rel_table <- data.frame(trait = traits,
r_sb = NA_real_, icc_3_k = NA_real_)
for (i in seq_along(traits)) {
rep <- run_reliability(sm[[traits[i]]],
n_permutations = 2000L,
seed = 1L, progress = FALSE)
rel_table$r_sb[i] <- rep$results$split_half$r_sb
rel_table$icc_3_k[i] <- rep$results$icc$icc_3_k
}
rel_tableLoaded from cache, the resulting table on this dataset:
| Trait | r_sb | ICC(3,1) | ICC(3,k) |
|---|---|---|---|
| competent | 0.31 | 0.02 | 0.30 |
| dominant | 0.52 | 0.05 | 0.51 |
| friendly | 0.82 | 0.18 | 0.81 |
| incompetent | 0.20 | 0.01 | 0.19 |
| intelligent | 0.55 | 0.06 | 0.54 |
| submissive | 0.30 | 0.02 | 0.29 |
| trust | 0.69 | 0.10 | 0.69 |
| unfriendly | 0.75 | 0.13 | 0.74 |
| unintelligent | 0.25 | 0.02 | 0.24 |
| untrust | 0.62 | 0.07 | 0.61 |
Spearman-Brown projected reliabilities and ICC(3,k) values are high throughout, indicating that the group-level CIs are stable across producer halves.
Cross-trait CI correlations
Restricted to the four traits used in the example contrasts (trust, friendly, competent, dominant), pairwise Pearson correlations between the group-mean CIs follow the expected valence structure: prosocial pairs correlate positively (trust–friendly +0.68, friendly–competent +0.48, trust–competent +0.42), and pairs that cross into dominance correlate negatively (friendly–dominant −0.51, trust–dominant −0.37). Competent–dominant is weakly negative (−0.22).
Correlations are computed over the pixels inside the parametric oval
face mask (make_face_mask(c(256L, 256L), region = "full")),
matching the masking step described in Oliveira et al. (2019, p. 892).
Without the mask, off-face pixels carry sinusoid energy from the noise
pool and attenuate every pair by roughly 0.02–0.05. The full ten-trait
matrix reproduces Oliveira et al. (2019) Table 2 to within ±0.02 across
all spot-checked cells.
| trust | friendly | competent | dominant | |
|---|---|---|---|---|
| trust | +0.68 | +0.42 | -0.37 | |
| friendly | +0.48 | -0.50 | ||
| competent | -0.22 | |||
| dominant |
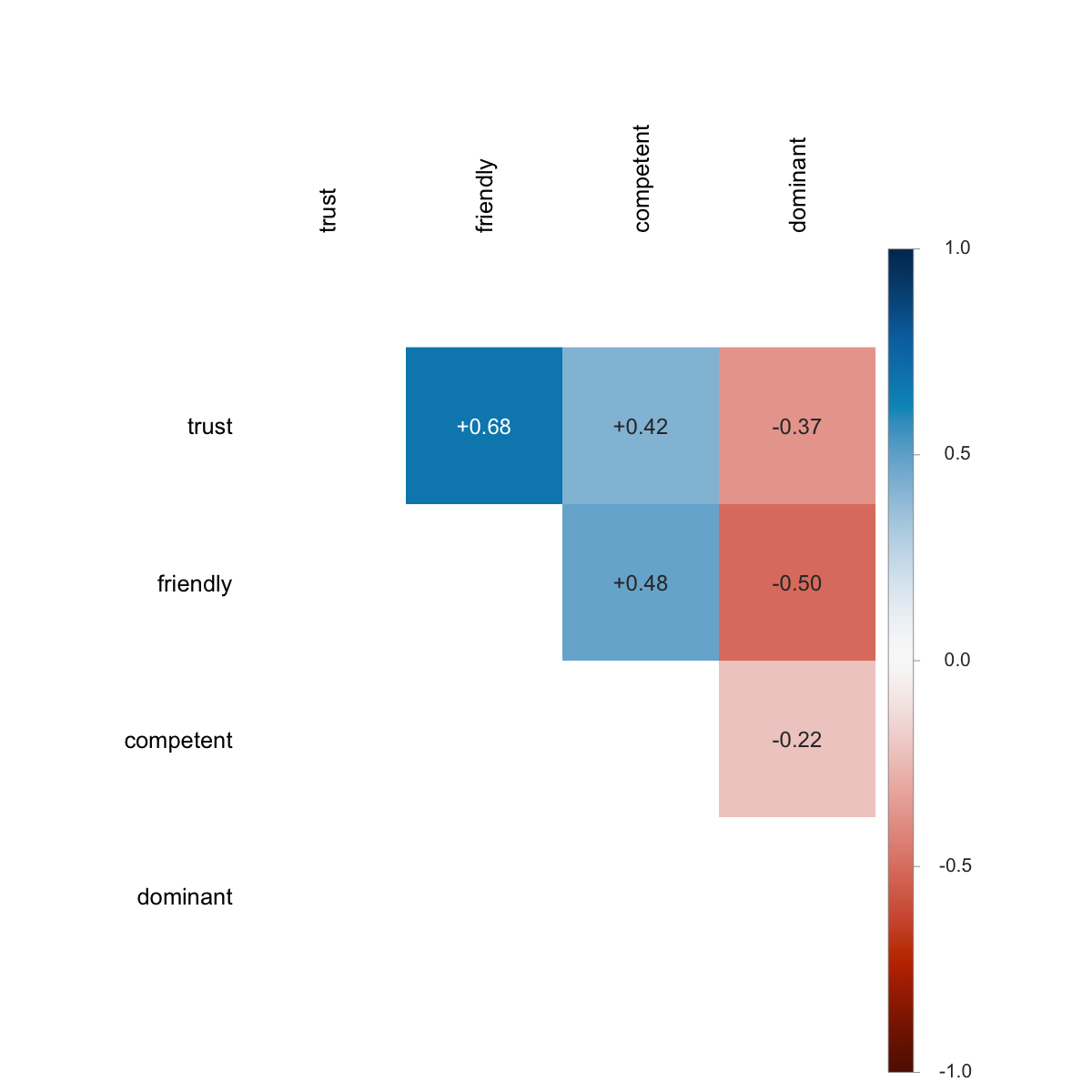
Pairwise Pearson correlations between the four group-mean CIs (trust, friendly, competent, dominant) in the Oliveira et al. (2019) Study 1 data. Diverging palette; blue = positive, red = negative; saturation indicates magnitude. Lower triangle and diagonal omitted because they are redundant.
ICC and group-mean infoVal z track each other across traits
Per-trait reliability and per-trait group-mean infoVal z (see §9) are tightly coupled across the ten conditions in this dataset: Pearson r = 0.97, 95% CI [0.88, 0.99], t(8) = 11.41, p < 0.001. Both quantify producer alignment on the pixel-level signal: ICC(3,k) as a variance ratio, group-mean z as the magnitude of the surviving group-mean signal against a matched reference. Conditions where producers converge on a common spatial template (friendly, unfriendly, trust) sit in the upper right; conditions where producer templates are largely idiosyncratic (incompetent, unintelligent) sit in the lower left.
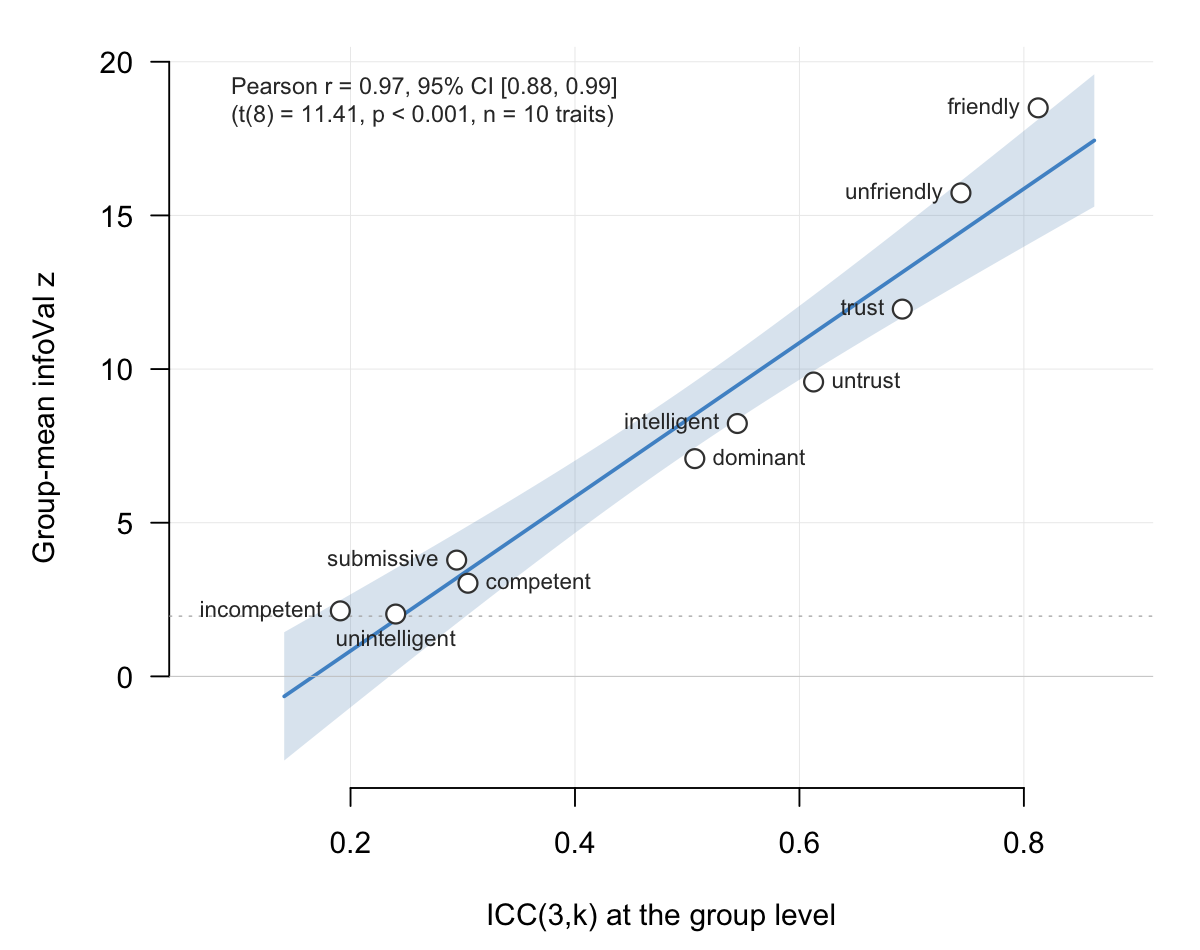
Per-trait ICC(3,k) versus per-trait group-mean infoVal z on the Oliveira et al. (2019) Study 1 data. Each point is one of the ten trait conditions; the line is the OLS fit and the band is its 95% confidence band. The dotted reference line marks group-mean z = 1.96.
12.7 Multi-contrast discriminability (full face)
Three motivating questions you can put to this dataset, going beyond the original paper:
- Trust vs Friendly: two trait labels often grouped under “warmth/morality”. Where do their visual representations diverge?
- Competent vs Dominant: two trait labels conceptually related to ability and agency but with opposite valence (Oliveira et al. 2019). Where on the face do they diverge?
- Trust vs Dominant: a cross-quadrant contrast spanning two functional dimensions, included as a reference benchmark.
Each contrast is a stratified cluster permutation test on the full
face. We summarise the overall magnitude of each divergence with
rel_dissimilarity() and lay them out side-by-side:
contrasts <- list(
"Trust vs Friendly" = list(a = sm[["trust"]], b = sm[["friendly"]]),
"Competent vs Dominant" = list(a = sm[["competent"]], b = sm[["dominant"]]),
"Trust vs Dominant" = list(a = sm[["trust"]], b = sm[["dominant"]])
)
# Full-face cluster tests, one per contrast.
ct_full <- lapply(contrasts, function(p) {
rel_cluster_test(p$a, p$b,
img_dims = c(256L, 256L),
cluster_threshold = 2.0,
n_permutations = 2000L,
seed = 1L,
progress = FALSE)
})
# Per-contrast Euclidean dissimilarity with bootstrap CIs.
dissim_full <- lapply(contrasts, function(p) {
rel_dissimilarity(p$a, p$b, n_boot = 2000L, seed = 1L,
progress = FALSE)
})
plot_dissimilarity_grid(
"Trust vs Friendly" = dissim_full[["Trust vs Friendly"]],
"Competent vs Dominant" = dissim_full[["Competent vs Dominant"]],
"Trust vs Dominant" = dissim_full[["Trust vs Dominant"]]
)Loaded from cache, the dissimilarity grid on this dataset:
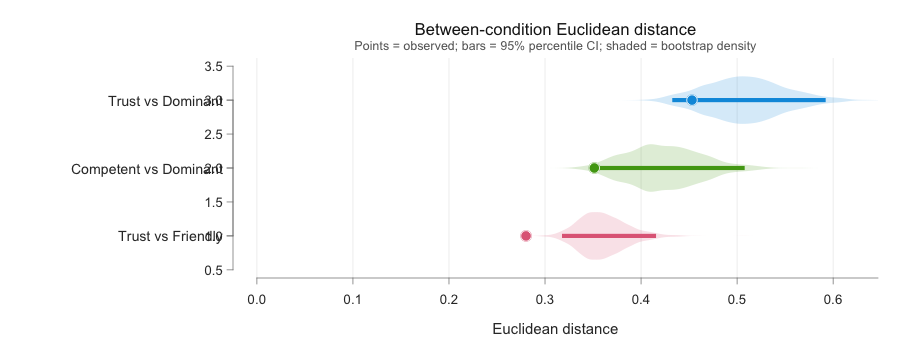
Between-condition Euclidean distance for the three contrasts on the Oliveira et al. (2019) Study 1 data. Each row is one contrast. The white-bordered point is the observed Euclidean distance between the two group-mean CIs, computed across all 65,536 pixels of the 256 x 256 image. The bar around it is the 95% percentile bootstrap CI from 2000 producer-level resamples (each condition resampled independently with replacement, distance recomputed on the resample). The shaded silhouette is the kernel density of the bootstrap distribution, scaled to the row height for visual comparison; its width does not encode units. Larger values mean the two group CIs sit farther apart in pixel space; bars whose left end is well above zero indicate that the separation is robust to producer-level variability.
The numeric values can be inspected on the dissimilarity objects themselves; the figure shows that all three contrasts diverge clearly on this dataset, with the lower bound of the 95% percentile CI well above zero.
12.8 Region-by-region cluster tests
A typical follow-up question is whether the divergences are uniform across the face or driven by specific anatomical regions. Run the cluster test once per region per contrast. The same three contrasts × four regions (full, eyes, mouth, upper face) gives twelve cells, which is small enough to scan as a table:
regions <- c("full", "eyes", "mouth", "upper_face")
cluster_grid <- expand.grid(
contrast = names(contrasts),
region = regions,
stringsAsFactors = FALSE
)
cluster_grid$n_clusters <- NA_integer_
cluster_grid$n_significant <- NA_integer_
cluster_grid$min_p <- NA_real_
for (i in seq_len(nrow(cluster_grid))) {
cname <- cluster_grid$contrast[i]
region <- cluster_grid$region[i]
m <- make_face_mask(c(256L, 256L), region = region)
ct <- rel_cluster_test(
contrasts[[cname]]$a, contrasts[[cname]]$b,
img_dims = c(256L, 256L),
mask = m,
cluster_threshold = 2.0,
n_permutations = 2000L,
seed = 1L,
progress = FALSE
)
cl <- ct$clusters
cluster_grid$n_clusters[i] <- if (is.null(cl)) 0L else
nrow(cl)
cluster_grid$n_significant[i] <-
sum(cl$significant, na.rm = TRUE)
cluster_grid$min_p[i] <-
if (is.null(cl) || nrow(cl) == 0L) NA_real_ else
min(cl$p_value, na.rm = TRUE)
}
cluster_gridLoaded from cache, the resulting grid on this dataset:
| Contrast | Region | n clusters | n significant | min p |
|---|---|---|---|---|
| Trust vs Friendly | full | 220 | 1 | 0.0430 |
| Competent vs Dominant | full | 223 | 3 | 0.0045 |
| Trust vs Dominant | full | 243 | 6 | 0.0000 |
| Trust vs Friendly | eyes | 83 | 0 | 0.2450 |
| Competent vs Dominant | eyes | 82 | 2 | 0.0150 |
| Trust vs Dominant | eyes | 101 | 4 | 0.0010 |
| Trust vs Friendly | mouth | 9 | 0 | 0.0630 |
| Competent vs Dominant | mouth | 10 | 2 | 0.0065 |
| Trust vs Dominant | mouth | 10 | 1 | 0.0200 |
| Trust vs Friendly | upper_face | 118 | 0 | 0.3115 |
| Competent vs Dominant | upper_face | 115 | 1 | 0.0185 |
| Trust vs Dominant | upper_face | 150 | 5 | 0.0050 |
The pattern of significant clusters across regions tells you where on the face each pair of conditions diverges and how strongly the producer sample agreed on those divergences (via FWER-controlled permutation). When a contrast shows a large full-face cluster but no significant clusters in any single region, the divergence is broad rather than localised; when the opposite holds, you have evidence for a localised contrast driven by one anatomical area.
12.9 Per-region informational value
Per-producer informational value also varies by region. A trait whose
group CI looks weak overall may carry stronger signal in one specific
region, and vice versa. Run infoval() per region per
condition:
# Helper: for one trait label, return a named integer vector
# giving the trial count per producer.
trial_counts_for <- function(label) {
trials <- raw[raw$trait == label, ]
ids <- unique(trials$participant_id)
counts <- as.integer(table(trials$participant_id)[ids])
names(counts) <- ids
counts
}
iv_grid <- expand.grid(
trait = c("trust", "friendly", "competent", "dominant"),
region = regions,
stringsAsFactors = FALSE
)
iv_grid$median_z <- NA_real_
iv_grid$n_above <- NA_integer_
for (i in seq_len(nrow(iv_grid))) {
label <- iv_grid$trait[i]
region <- iv_grid$region[i]
sm <- get(paste0("sm_", label))
tc <- trial_counts_for(label)
m <- make_face_mask(c(256L, 256L), region = region)
iv <- infoval(sm, noise_matrix, tc,
iter = 1000L,
mask = m,
seed = 1L,
progress = FALSE)
iv_grid$median_z[i] <- stats::median(iv$infoval)
iv_grid$n_above[i] <- sum(iv$infoval >= 1.96)
}
iv_gridLoaded from cache, the resulting grid on this dataset:
| Trait | Region | Median producer z | n above 1.96 (of 20) |
|---|---|---|---|
| trust | full | +0.50 | 3 |
| friendly | full | +0.97 | 5 |
| competent | full | +0.70 | 3 |
| dominant | full | +0.89 | 6 |
| trust | eyes | +0.50 | 1 |
| friendly | eyes | +0.34 | 2 |
| competent | eyes | +0.36 | 2 |
| dominant | eyes | +0.91 | 3 |
| trust | mouth | +0.53 | 3 |
| friendly | mouth | +0.75 | 4 |
| competent | mouth | +0.25 | 5 |
| dominant | mouth | +0.38 | 2 |
| trust | upper_face | +0.30 | 2 |
| friendly | upper_face | +0.23 | 2 |
| competent | upper_face | +0.21 | 3 |
| dominant | upper_face | +0.37 | 5 |
Compared with the full-face infoVal table in §12.4, this region-restricted view often shifts the picture. A trait whose group CI looks weak overall may carry stronger signal in one specific region, and a trait that looks strong overall may localise to one region rather than spanning the whole face.
In this dataset the regional pattern is informative on its own. Dominance carries comparatively strong signal in the eyes (median producer z noticeably higher than the other three traits in that region) and weaker, comparable signal in the mouth and upper face. Friendliness flips that pattern, with the mouth carrying its strongest regional signal and the eyes its weakest. Trustworthy localises broadly across the eyes and mouth without a clear regional peak, and competent’s signal is the most evenly spread across regions, though at uniformly modest levels. The headline full-face median for each trait masks these regional contrasts.
The two grids (the cluster grid from §12.8 and the infoVal grid here) answer two different questions about the same masked region. The cluster test asks where conditions A and B disagree; infoVal asks how informative a single condition’s mask is when restricted to this region. Reporting both side-by-side gives a fuller picture of how producers’ representations organise across the face.
12.10 Pairwise cluster maps for two motivating contrasts
The two contrasts highlighted in this section are chosen because they sit at opposite ends of a methodological prediction. Trust versus Friendly pits two traits that load on the same warmth dimension of social judgement and share much of their facial encoding, so the expectation is a comparatively narrow set of pixel-level differences. Dominant versus Competent pits two traits that the original paper (Oliveira et al., 2019) argues dissociate: dominance is read off coarser whole-face structure while competence draws on finer ability cues, and the two should therefore diverge over a broader spatial region. The maps below let the reader judge whether the data agree.
For each between-condition contrast we render two complementary maps on the same male base face. The descriptive map shows the difference of the two group-mean CIs across all pixels in the face oval, with no inferential filter applied. It lets the reader see the raw spatial pattern of agreement first, before any statistical thresholding decisions are layered on. The FWER-controlled map shows the same difference, but restricted to pixels that fall inside a cluster significant at p < .05 under FWER-controlled cluster-based permutation testing.
The descriptive map answers “where do the two group CIs diverge, descriptively?”. The FWER-controlled map answers “which of those divergences are large enough that we should believe they reflect a real between-condition difference rather than noise?”. Showing both side-by-side lets the reader see how much of the descriptive pattern is supported by the data once a controlled Type I error filter is applied.
For each contrast we (i) take the difference of the two group- mean
CIs, (ii) render it on the base face (descriptive view), (iii) run
rel_cluster_test() to find spatially contiguous regions
where the per-pixel Welch t exceeds the cluster-forming threshold, and
(iv) overlay the same difference on the base face restricted to pixels
belonging to a significant cluster (2000 stratified label permutations,
max-mass null, cluster threshold |t| > 2.0).
# Descriptive map: difference of group means, no significance filter.
diff_signal <- rowMeans(sm[["trust"]]) - rowMeans(sm[["friendly"]])
plot_ci_overlay(
diff_signal,
base_image = "base.jpg",
mask = make_face_mask(c(256L, 256L), region = "full"),
main = "Trust minus Friendly (descriptive)"
)
# FWER-controlled map: same difference, masked to significant clusters.
ct_tf <- rel_cluster_test(
sm[["trust"]], sm[["friendly"]],
img_dims = c(256L, 256L),
cluster_threshold = 2.0,
n_permutations = 2000L,
seed = 1L
)
# Pull out the cluster ids of the significant clusters in each
# direction (Trust > Friendly = "pos", Friendly > Trust = "neg").
sig_pos <- ct_tf$clusters$cluster_id[ct_tf$clusters$direction == "pos" &
ct_tf$clusters$significant]
sig_neg <- ct_tf$clusters$cluster_id[ct_tf$clusters$direction == "neg" &
ct_tf$clusters$significant]
# Build a logical mask marking pixels that fall inside any
# significant cluster (in either direction). `pos_labels` and
# `neg_labels` are integer matrices of cluster ids; %in% checks
# membership pixel by pixel.
sig_mask <- (ct_tf$pos_labels %in% sig_pos) |
(ct_tf$neg_labels %in% sig_neg)
# Plot the difference (Trust minus Friendly) on the base face.
diff_signal <- rowMeans(sm[["trust"]]) - rowMeans(sm[["friendly"]])
plot_ci_overlay(
diff_signal,
base_image = "base.jpg",
mask = as.vector(sig_mask),
main = "Trust minus Friendly (FWER-controlled clusters)"
)Descriptive maps. No significance filter applied; the display is restricted to the face oval so the colour scale is not dominated by hair/background pixels.


Descriptive pairwise difference maps on the male base face. Left: Trust minus Friendly. Right: Dominant minus Competent. Blue = first condition stronger; red = second condition stronger; opacity = magnitude of the difference. The display covers every pixel in the full-face oval; no inferential filter is applied. These maps show the raw spatial pattern of agreement before any cluster-based permutation testing.
FWER-controlled maps. Same difference signals, but pixels outside any significant cluster appear transparent so the base face shows through.


FWER-controlled pairwise cluster-agreement maps on the male base face. Left: Trust minus Friendly. Right: Dominant minus Competent. Each map shows the difference of the two group-mean CIs only at pixels belonging to a cluster that is significant at p < .05 under FWER-controlled cluster-based permutation testing (cluster threshold |t| > 2.0; 2000 stratified label permutations; max-mass null). Colour convention as in the descriptive maps above (blue = first condition stronger; red = second condition stronger; opacity = magnitude). Compare with the descriptive maps to see how much of the raw pattern survives the inferential filter.
The two contrasts pick out qualitatively different spatial signatures, broadly consistent with the prediction set out at the start of this section. Trust versus Friendly localises around the eye and mid-face regions, consistent with the warmth dimension being read off socially-relevant features and shared across the two traits. Dominant versus Competent spreads more widely across the face, consistent with a contrast that draws on both whole-face agency cues and finer ability cues. Among the pixels that survive the FWER filter, the Dominant vs Competent map retains noticeably more spatial extent than the Trust vs Friendly map. These maps extend Oliveira et al. (2019) by adding a between-condition inferential filter the original paper did not run.
13. Brief-RC end-to-end
The Brief-RC workflow follows the same diagnose, compute, and assess
flow with two practical differences. First, the response data has
multiple alternatives per trial (12 or 20, recorded as one row per trial
carrying the chosen pool id and sign). Second, the noise matrix is
consumed directly, without an rcicr wrapper.
13.1 Brief-RC variants currently supported
ci_from_responses_briefrc() accepts two
method values, both matching the variants validated in
Schmitz, Rougier, & Yzerbyt (2024):
-
"briefrc12"(default): 12 alternatives per trial, 6 original + 6 inverted, arranged in a 3 x 4 grid in the Schmitz et al. study. -
"briefrc20": 20 alternatives per trial, 10 original + 10 inverted, arranged in a 4 x 5 grid in the Schmitz et al. study.
The CI computation is identical for both variants. Schmitz’s
genMask() formula does not depend on how many alternatives
are shown per trial; it always reduces to mean-by-stim of the chosen
pool ids, divided by length(unique(chosen_stim)). The
package’s random-responder reference simulator for
infoval() similarly relies on a 50/50 original/inverted
marginal per trial, which holds for any symmetric Brief-RC split. The
method argument is therefore metadata: it is recorded in
the result list (as $method) so that downstream code,
reports, and citations can indicate which paradigm produced the data,
but it does not branch the computation.
Other split sizes (Brief-RC 4, 6, 8, 10) are mentioned in the Schmitz
et al. (2024) discussion as possible future variants worth
investigating, but they have not been validated and are not exposed in
rcisignal. If you genuinely need to compute masks for one
of those configurations, the §12.5 hand-rolled five-step recipe applies
unchanged (the formula is symmetric in the per-trial split), but the
result should be reported as exploratory.
13.2 End-to-end Brief-RC example
library(rcisignal)
# 1. Read the Schmitz et al. 2024 noise matrix directly. You
# can also generate your own pool with rcicr (one-off,
# slow); read_noise_matrix() handles both.
nm <- read_noise_matrix("schmitz/noise_matrix.txt")
# 2. Diagnostics on Brief-RC responses.
report <- run_diagnostics(
briefrc_responses,
method = "briefrc",
noise_matrix = nm,
expected_n = 60L,
baseimage = "base.jpg",
infoval_iter = 1000L
)
report
# 3. Compute individual masks.
res <- ci_from_responses_briefrc(
briefrc_responses,
noise_matrix = nm,
method = "briefrc12",
scaling = "none" # base_image not needed at scaling = "none"
)
signal <- res$signal_matrix
# 4. Reliability assessment (same metrics, same calls).
run_reliability(signal, seed = 1L)
# 5. Per-producer infoVal with trial-count-matched reference.
infoval(signal, nm,
trial_counts = setNames(rep(60L, ncol(signal)),
colnames(signal)),
iter = 1000L, seed = 1L)
# 6. Save rendered CIs to PNG (visualisation only). Do not
# feed these to rel_* or to hand-rolled infoVal.
res_render <- ci_from_responses_briefrc(
briefrc_responses,
noise_matrix = nm,
base_image = "base.jpg", # required when scaling != "none"
scaling = "matched" # Schmitz Experiment 2 convention
)
# res_render$rendered_ci is base + matched(mask), ready for PNG14. Caveats and reporting notes
A summary of what to keep in mind when reporting results.
Reliability and validity address different
questions. The metrics in this package quantify whether a CI is
stable (within-condition) and separable (between-condition). Whether the
CI accurately reflects the producer’s mental representation of the
target trait is a separate validity question, typically addressed by an
external rater study or a behavioural validation, and the package does
not address it. High rel_* values support claims about
consistency and discriminability; plan validity work alongside the
rcisignal pipeline.
Raw vs rendered. Pre-rendered PNGs are convenient
and carry the scaling step into your pixel data. Variance-based metrics
break under any scaling; correlation-based metrics survive a single
uniform scaling and break under per-CI “matched” scaling. The package
errors at runtime when a known-rendered matrix is fed to a
variance-based metric. The cleanest workflow computes CIs from raw
responses (ci_from_responses_*), feeds the returned
$signal_matrix to all metrics, and renders to PNG only for
visualisation.
Group-mean z and per-producer z carry different information. Per-producer Frobenius norms aggregate over the whole image and dilute localised signal, so individual z values are systematically lower than group-mean z even when the group CI is highly informative (the §12.4 pattern). Report both, framed as different-grain statistics.
FWER scope. rel_cluster_test() controls
FWER across pixels within a single comparison.
run_discriminability_pairwise() adds a second layer of FWER
control across the K-choose-2 pair comparisons (Holm by default). Don’t
double-correct: within-pair cluster p-values are already adjusted; the
across-pairs Holm operates on the per-pair minimum cluster p.
Apply masks symmetrically. When
infoval() uses a mask, both the observed Frobenius norm and
the reference distribution are restricted to the same pixels. Other
functions follow the same discipline. Mixing masked observed with
unmasked reference (or vice versa) yields a number with no defensible
interpretation.
Sample size. Reliability estimates themselves become unreliable below N approximately 30 per condition. The package warns at N < 30 and aborts at N < 4. Aim for N >= 60 per condition for stable assessment.
Pre-1.0 status. The package is not yet at version 1.0; argument names and defaults may change between minor versions, particularly when the change makes a sharp edge less easy to cut yourself on. NEWS.md documents every breaking change.
15. Appendix: troubleshooting low or negative infoVal
This appendix expands on the brief interpretation note in §9. If you
compute infoval() and find that most or
all per-producer z-scores sit well below 1.96, sometimes
negative, even though spot checks suggest producers are doing the task
seriously, that is a common pattern rather than evidence of a data
problem. Five reasons in roughly the order they tend to apply.
Frobenius norm is a global energy statistic. It sums squared pixel deviations across the entire image. Real internal representations are usually spatially sparse, concentrated in face features (eyes, mouth, jaw) rather than spanning the whole image grid. Pixels outside that region contribute noise of similar magnitude to the chance reference, and that noise dilutes the signal-bearing region in the global norm. A producer with strong, visually-obvious signal in the eyes can therefore have a Frobenius norm only marginally above the random reference. (See point 4 below for the practical countermeasure.)
The reference is strict because it lives in the same subspace. Both the observed mask and the reference are projections onto the same low-dimensional sinusoidal noise basis. The reference distribution has plenty of overall energy by construction, so the only way to clear z = 1.96 is to align signs with a specific subset of patterns more than chance.
Per-trial signal is small. Each 2IFC choice contributes a tiny signal increment relative to the per-trial noise amplitude. With 300 trials the SNR gain is sqrt(300) ~ 17x, but if per-trial signal is on the order of 5% of per-trial noise, post-aggregation effective SNR is barely visible to a global energy measure.
Without a face mask, infoVal counts background.
make_face_mask()ships a parametric full-face oval that isolates the face region from background and hair. Applying it (infoval(..., mask = make_face_mask(c(256, 256)))) concentrates the norm on signal-bearing pixels and typically lifts z-scores noticeably. The convention of applying a full-face oval before computing pixel-wise CI metrics follows prior practice in social-face RC (e.g., Oliveira et al., 2019; Ratner et al., 2014; Schmitz, Rougier, & Yzerbyt, 2024).-
Group-level CIs can show higher z than individual CIs, but the inflation is conditional, not structural. Two pieces of math feed into this, and both need stating. First, averaging
Nproducer masks under independent-noise assumptions reduces noise variance by1/Nand the noise norm by1/sqrt(N). So the group-mean CI ofNproducers each contributingTtrials has noise comparable to a single producer withN x Ttrials (a 20-producer condition at 300 trials each gives an effective6000-trial noise floor). Second, whether that noise reduction actually translates into a sqrt(N)-style increase in group-mean infoVal over the per-producer median depends on whether the producers’ signals are spatially aligned. If producers share a common spatial template, signal survives the averaging and the group-mean z grows roughly as sqrt(N) above the per-producer floor. If producer templates are idiosyncratic, averaging cancels signal as well as noise and the group-mean z stays modest. The Oliveira et al. (2019) reanalysis in §12.4 shows both regimes in the same dataset: friendly / unfriendly, where per-producer signal is broadly shared, land at group-mean z of roughly 15-18; incompetent / unintelligent, where alignment is weaker, sit near z = 2. The ratiogroup_z / per_producer_medianis therefore not a fixed inflation factor; it is a joint readout ofN,T, and producer alignment.Brinkman et al. (2019) only ever computed infoVal on individual CIs. They reported a mean per-producer infoVal of 3.9 (lab) and 2.9 (online), with 68% / 54% of producers individually exceeding 1.96 (their pp. 12, 14). For group- level reporting they explicitly recommended inspecting the distribution of per-producer infoVals contributing to the group CI rather than computing a single infoVal on the averaged noise pattern (their p. 13). The group-mean infoVal this package offers (
group_mean_z(), called insidediagnose_infoval()) is a package-level extension of that recipe and has not been independently validated for social- face RC (see §1.2). The §12.4 worked example reports both numbers alongside; treat the group-mean z as a supplementary headline, not a replacement for the Brinkman-style per- producer distribution.
15.1 Diagnostic recipe
If a per-producer infoVal table looks worryingly low, work through these steps before reporting it:
sm <- res$signal_matrix
tc <- setNames(rep(300L, ncol(sm)), colnames(sm))
# 1. Compare observed and reference norm distributions directly.
iv <- infoval(sm, noise_matrix, tc, iter = 1000L, seed = 1L)
ref <- iv$reference[[as.character(tc[1])]]
cat(sprintf(
"observed median = %.4f, reference median = %.4f, %% above = %+.1f%%\n",
median(iv$norms), median(ref),
100 * (median(iv$norms) - median(ref)) / median(ref)
))
# 2. Apply the face mask. Per-producer z usually rises.
fm <- make_face_mask(c(256L, 256L))
iv_masked <- infoval(sm, noise_matrix, tc, mask = fm,
iter = 1000L, seed = 1L)
median(iv_masked$infoval)
# 3. Compute the group-mean CI's infoVal.
group <- matrix(rowMeans(sm), ncol = 1,
dimnames = list(NULL, "group"))
tc_grp <- setNames(sum(tc), "group")
iv_grp <- infoval(group, noise_matrix, tc_grp,
iter = 1000L, seed = 1L)
iv_grp$infoval # value depends on signal alignment;
# see paragraph 5 of §15
# 4. Sanity-check the chance baseline. A random-mask producer
# should give z ~ 0 within MAD noise.
random_mask <- (noise_matrix[, sample(ncol(noise_matrix),
300L, replace = TRUE)] %*%
sample(c(-1, 1), 300L, replace = TRUE)) / 300
iv_rand <- infoval(matrix(random_mask, ncol = 1,
dimnames = list(NULL, "rnd")),
noise_matrix, setNames(300L, "rnd"),
iter = 1000L, seed = 1L)
iv_rand$infoval # should be ~ 0 within MAD noise15.2 What clearly negative z-scores mean
A negative z indicates that the observed mask carries less Frobenius
energy than the chance reference. This is informative rather than a
calibration error. A clearly negative z (say, below -2) on a producer
who allegedly engaged with the task suggests they responded
inconsistently, partly randomly, or with selection patterns that average
toward zero. Cross-check rel_loo_z(), response-time
distributions, and any other attention checks before drawing
conclusions.
15.3 What to report
For a publishable summary we typically recommend two complementary statistics:
- The median per-producer infoVal z and the proportion of producers above z = 1.96, mirroring Brinkman et al.’s (2019) reporting choice.
- The group-mean CI’s infoVal z as a supplementary headline number. Under signal alignment this can be substantially larger than the per-producer median, with a sqrt(N)-style upper envelope; under weak alignment it stays close to per-producer values (paragraph 5 of §15 spells out why). This metric is a package-level extension and has not been independently validated for social-face RC (§1.2), so report it as exploratory rather than primary.
The two numbers answer different questions. The median tells you how informative a typical individual CI is; the group-mean z tells you how informative the condition’s average CI is.
16. Citation
if (requireNamespace("rcisignal", quietly = TRUE)) {
print(citation("rcisignal"))
} else {
message(
"Install rcisignal to view its citation: ",
"devtools::install() or ",
"remotes::install_github(\"olivethree/rcisignal\")."
)
}
#> To cite package 'rcisignal' in publications use:
#>
#> Oliveira M (2026). _rcisignal: Quality Checks for Reverse-Correlation
#> Data and Classification Images_. R package version 0.1.5,
#> <https://github.com/olivethree/rcisignal>.
#>
#> A BibTeX entry for LaTeX users is
#>
#> @Manual{,
#> title = {rcisignal: Quality Checks for Reverse-Correlation Data and Classification
#> Images},
#> author = {Manuel Oliveira},
#> year = {2026},
#> note = {R package version 0.1.5},
#> url = {https://github.com/olivethree/rcisignal},
#> }17. References
Brinkman, L., Goffin, S., van de Schoot, R., van Haren, N. E. M., Dotsch, R., & Aarts, H. (2019). Quantifying the informational value of classification images. Behavior Research Methods, 51(5), 2059-2073. https://doi.org/10.3758/s13428-019-01232-2
Brinkman, L., Todorov, A., & Dotsch, R. (2017). Visualising mental representations: A primer on noise-based reverse correlation in social psychology. European Review of Social Psychology, 28(1), 333-361. https://doi.org/10.1080/10463283.2017.1381469
Brown, W. (1910). Some experimental results in the correlation of mental abilities. British Journal of Psychology, 3(3), 296-322. https://doi.org/10.1111/j.2044-8295.1910.tb00207.x
Chauvin, A., Worsley, K. J., Schyns, P. G., Arguin, M., & Gosselin, F. (2005). Accurate statistical tests for smooth classification images. Journal of Vision, 5(9), 1. https://doi.org/10.1167/5.9.1
Cone, J., Brown-Iannuzzi, J. L., Lei, R., & Dotsch, R. (2021). Type I error is inflated in the two-phase reverse correlation procedure. Social Psychological and Personality Science, 12(5), 760-768. https://doi.org/10.1177/1948550620938616
Cook, R. D. (1977). Detection of influential observation in linear regression. Technometrics, 19(1), 15-18. https://doi.org/10.1080/00401706.1977.10489493
DeBruine, L. (2022). webmorphR: Reproducible stimuli. https://github.com/debruine/webmorphR
Dotsch, R. (2016, 2023). rcicr: Reverse-Correlation Image- Classification Toolbox. https://github.com/rdotsch/rcicr
Koo, T. K., & Li, M. Y. (2016). A guideline of selecting and reporting intraclass correlation coefficients for reliability research. Journal of Chiropractic Medicine, 15(2), 155-163. https://doi.org/10.1016/j.jcm.2016.02.012
Maris, E., & Oostenveld, R. (2007). Nonparametric statistical testing of EEG- and MEG-data. Journal of Neuroscience Methods, 164(1), 177-190. https://doi.org/10.1016/j.jneumeth.2007.03.024
McGraw, K. O., & Wong, S. P. (1996). Forming inferences about some intraclass correlation coefficients. Psychological Methods, 1(1), 30-46. https://doi.org/10.1037/1082-989X.1.1.30
Nichols, T. E., & Holmes, A. P. (2002). Nonparametric permutation tests for functional neuroimaging: a primer with examples. Human Brain Mapping, 15(1), 1-25. https://doi.org/10.1002/hbm.1058
Oliveira, M., Garcia-Marques, T., Dotsch, R., & Garcia-Marques, L. (2019). Dominance and competence face to face: Dissociations obtained with a reverse correlation approach. European Journal of Social Psychology. https://doi.org/10.1002/ejsp.2569
Ratner, K. G., Dotsch, R., Wigboldus, D. H. J., van Knippenberg, A., & Amodio, D. M. (2014). Visualizing minimal ingroup and outgroup faces: implications for impressions, attitudes, and behavior. Journal of Personality and Social Psychology, 106(6), 897-911. https://doi.org/10.1037/a0036498
Schmitz, M., Rougier, M., & Yzerbyt, V. (2020). Comment on “Quantifying the informational value of classification images”: A miscomputation of the infoVal metric. Behavior Research Methods, 52(3), 1383-1386. https://doi.org/10.3758/s13428-019-01295-1
Schmitz, M., Rougier, M., Yzerbyt, V., Brinkman, L., & Dotsch, R. (2020). Erratum to: Comment on “Quantifying the informational value of classification images”: Miscomputation of infoVal metric was a minor issue and is now corrected. Behavior Research Methods, 52(4), 1800-1801. https://doi.org/10.3758/s13428-020-01367-7
Schmitz, M., Rougier, M., & Yzerbyt, V. (2024). Introducing the brief reverse correlation: an improved tool to assess visual representations. European Journal of Social Psychology. https://doi.org/10.1002/ejsp.3100
Shrout, P. E., & Fleiss, J. L. (1979). Intraclass correlations: uses in assessing rater reliability. Psychological Bulletin, 86(2), 420-428. https://doi.org/10.1037/0033-2909.86.2.420
Smith, S. M., & Nichols, T. E. (2009). Threshold-free cluster enhancement: addressing problems of smoothing, threshold dependence and localisation in cluster inference. NeuroImage, 44(1), 83-98. https://doi.org/10.1016/j.neuroimage.2008.03.061
Spearman, C. (1910). Correlation calculated from faulty data. British Journal of Psychology, 3(3), 271-295. https://doi.org/10.1111/j.2044-8295.1910.tb00206.x
Tukey, J. W. (1958). Bias and confidence in not-quite large samples [Abstract]. Annals of Mathematical Statistics, 29(2), 614. https://doi.org/10.1214/aoms/1177706647